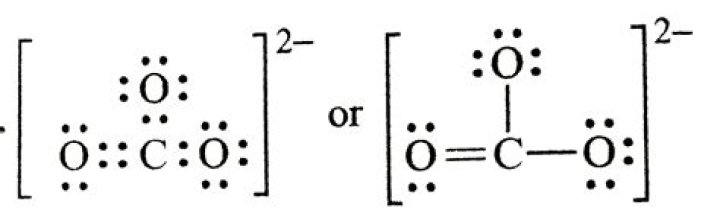
What is the Lewis structure for carbonate

Andrew White
Published Feb 12, 2026
The carbonate ion is the simplest oxocarbon anion, consisting of one carbon atom surrounded by three oxygen atoms in a trigonal planar arrangement. The Lewis structure of the carbonate ion has two single bonds to negative oxygen atoms and one short double bond to a neutral oxygen.
What is the structure of carbonate?
The carbonate ion is the simplest oxocarbon anion, consisting of one carbon atom surrounded by three oxygen atoms in a trigonal planar arrangement. The Lewis structure of the carbonate ion has two single bonds to negative oxygen atoms and one short double bond to a neutral oxygen.
How many Lewis structures can be drawn for co32?
There are three different possible resonance structures from carbonate. Each carbon oxygen bond can be thought of as 1.333 bonds. the average of a double bond and 2 single bonds.
What is the Lewis representation of CO3 2?
CO32- is an anion (a negative ion) seen frequently in chemistry. In the CO32- Lewis structure carbon is the least electronnegative element. Therefore it is put in the center of the dot structure. For the CO32- Lewis structure there are a total of 24 valence electrons available.How do you draw a Lewis structure?
- Step 1: Find the Total Number of Valence Electrons. …
- Step 2: Find the Number of Electrons Needed to Make the Atoms “Happy” …
- Step 3: Determine the Number of Bonds in the Molecule. …
- Step 4: Choose a Central Atom. …
- Step 5: Draw a Skeletal Structure. …
- Step 6: Place Electrons Around Outside Atoms.
What is the shape of CO3 2?
3 that the molecular geometry of CO 3 2− is trigonal planar with bond angles of 120°.
How is CO3 2 formed?
Lewis structure of CO32- (carbonate) ion In carbonate ion, there is two oxygen atoms which has -1 charge on each of them. One of these oxygen atom take a proton (H+ ion) and form a -OH group.
What is the formula for metal carbonate?
Molecular formula for metal carbonates is CO2−3 and can be considered as carbonate ester.How do you write carbonate?
Carbonate Ion is a polyatomic ion with formula of CO3(2-).
What is the shape of OF2?The molecular geometry of OF2 is bent and its electron geometry is tetrahedral because the presence of two lone pairs on the central atom creates repulsion with bonded pairs of electrons, as a result, all outer atoms(fluorine) pushes down in order to minimize the repulsion according to the VSEPR theory, and that makes …
Article first time published onDoes co32 have resonance structures?
Unlike O3, though, the actual structure of CO32− is an average of three resonance structures. … Carbon has 4 valence electrons, each oxygen has 6 valence electrons, and there are 2 more for the −2 charge. This gives 4 + (3 × 6) + 2 = 24 valence electrons.
How many electrons does co32 have?
The Carbonate (CO2−3) Ion Carbon has 4 valence electrons, each oxygen has 6 valence electrons, and there are 2 more for the −2 charge. This gives 4 + (3 × 6) + 2 = 24 valence electrons.
Is CO2 a resonance structure?
Carbon dioxide, or CO2 , has three resonance structures, out of which one is a major contributor. The CO2 molecule has a total of 16 valence electrons – 4 from carbon and 6 from each oxygen atom.
What do Lewis structures show?
A Lewis Structure is a very simplified representation of the valence shell electrons in a molecule. It is used to show how the electrons are arranged around individual atoms in a molecule. Electrons are shown as “dots” or for bonding electrons as a line between the two atoms.
How do Lewis structures work?
A Lewis electron dot diagram (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The number of dots equals the number of valence electrons in the atom. … The total number of electrons does not change.
What shape is h2o?
Water has 4 regions of electron density around the central oxygen atom (2 bonds and 2 lone pairs). These are arranged in a tetrahedral shape. The resulting molecular shape is bent with an H-O-H angle of 104.5°.
What type of bond is co3 2?
The carbonate anion is a covalent compound anion with a net charge of -2.
Is carbonate trigonal planar?
Carbonate Ion: The carbon and oxygen are bonded through a double bond which counts as “one electron pair” and two single bonded oxygens. Hence the molecule has three electron pairs and is trigonal planar geometry.
Is CO32 symmetrical?
It’s not, because of symmetry. For the same reason that , , and are all non-polar. The carbonate ion is resonance stabilized: In the resonance hybrid, all three oxygen atoms are identical; all three C—O bonds are identical.
What is the formal charge of C in co32 −?
Therefore the formal charge of the carbon atom is 0.
How do you write chemical formulas?
- In a chemical equation, the reactants are written on the left, and the products are written on the right.
- The coefficients next to the symbols of entities indicate the number of moles of a substance produced or used in the chemical reaction.
Are carbonates bases?
Carbonates are moderately strong bases. Aqueous solutions are basic because the carbonate anion can accept a hydrogen ion from water. CO32− + H2O ⇌ HCO3− + OH− Carbonates react with acids, forming salts of the metal, gaseous carbon dioxide, and water.
What elements make up carbonate?
Calcium carbonate is a chemical compound with the formula CaCO3 formed by three main elements: carbon, oxygen, and calcium.
What is the correct formula for sulfite?
PubChem CID1099StructureFind Similar StructuresMolecular FormulaO3S-2Synonymssulfite Sulfite ion Sulphite Sulfite ions 14265-45-3 More…Molecular Weight80.07



