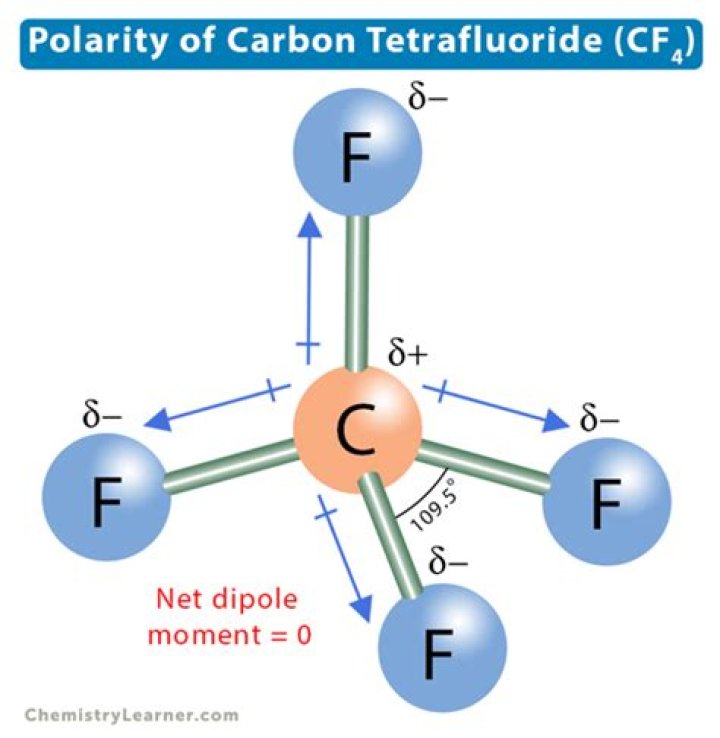
What is the mass of a carbon tetrafluoride molecule grams

Andrew Campbell
Published Feb 16, 2026
Property NameProperty ValueReferenceMonoisotopic Mass87.99361265Computed by PubChem 2.1 (PubChem release 2021.05.07)Topological Polar Surface Area0 ŲComputed by Cactvs 3.4.8.18 (PubChem release 2021.05.07)Heavy Atom Count5Computed by PubChemFormal Charge0Computed by PubChem
What is the mass of a carbon tetrafluoride molecule?
Namesshow SMILESPropertiesChemical formulaCF4Molar mass88.0043 g/mol
How many grams is a molecule of carbon?
12 atoms x 1.66 x 10-24g x 6.022 x 1023 atoms/ mol= 12 g/mol. So a single carbon atom weighs 12 amu while a mole of carbon atoms weighs 12.01g/mol.
What is the mass of a carbon tetrachloride molecule grams?
The molar mass of carbon is 12.011 g/mol. The molar mass of chlorine is 35.45 g/mol. The molar mass of carbon tetrachloride is 153.81 grams per mole.What is the molar mass of Dicarbon Tetrahydride?
You do the math. The molar mass of carbon tetrachloride is 153.8 g/mol.
How do you find the mass?
- mass=density×volume (m=ρV). Density is a measure of mass per unit of volume, so the mass of an object can be determined by multiplying density by volume.
- mass=force÷acceleration (m=F/a). …
- mass=weight÷gravitational acceleration (m=W/g).
How do you convert grams to mass?
Gram [g]Earth’s Mass50 g8.366800535475E-27 Earth’s mass100 g1.673360107095E-26 Earth’s mass1000 g1.673360107095E-25 Earth’s mass
How many grams are in a molecule?
This can also be written as 6.022×1023 mol-1. The mass of one mole of a substance is equal to that substance’s molecular weight. For example, the mean molecular weight of water is 18.015 atomic mass units (amu), so one mole of water weight 18.015 grams.What is mass formula?
Mass is defined as the amount of matter present in a body. The SI unit of mass is the kilogram (kg). The formula of mass can be written as: Mass = Density × Volume.
Why is a mole 6.022 x10 23?One mole is equal to 6.022×1023 units. A mole is an important unit because on the periodic table a mole of a substance is equal to its atomic mass in grams. … This means that 6.022×1023 carbon atoms (or molecules) weights 12.01 grams.
Article first time published onWhat is the formula for moles to grams?
FORMULAS Related LinksVariance And Standard Deviation FormulaEquation Of A Tangent To A Circle
What is the mass of a sulfur hexafluoride molecule?
NamesMolar mass146.06 g/molAppearanceColorless gasOdorodorlessDensity6.17 g/L
How do you write Disulfur tetrafluoride?
The formula of disulfur tetrachloride is S2F4.
How do you make sulfur tetrafluoride?
SF4 is produced by the reaction of SCl2 and NaF in acetonitrile: 3 SCl2 + 4 NaF → SF4 + S2Cl2 + 4 NaCl.
How do you write carbon tetrafluoride?
Carbon tetrafluoride | CF4 – PubChem.
How do you find the atomic mass of carbon in grams?
The atomic mass of carbon would be 12.01 grams per mole of carbon atoms. To calculate the atomic mass of a single atom of an element, add up the mass of protons and neutrons. Example: Find the atomic mass of an isotope of carbon that has 7 neutrons.
What is the mass of molecule of co2 in grams?
The molecular mass of carbon dioxide is 44.01amu. The molar mass of any compound is the mass in grams of one mole of that compound. One mole of carbon dioxide molecules has a mass of 44.01g, while one mole of sodium sulfide formula units has a mass of 78.04g.
Can you convert weight to mass?
Mass does not change with location. To find an object’s mass using its weight, the formula is Mass equals Weight divided by the Acceleration of Gravity (M = W ÷ G). Convert the weight measured in pounds to the equivalent in Newtons. In the formula for determining mass based on weight, mass is measured in Newtons.
Is gram a mass?
gram (g), also spelled gramme, unit of mass or weight that is used especially in the centimetre-gram-second system of measurement (see International System of Units). … The gram is now defined as 0.001 kilogram (kg), which is defined in terms of Planck’s constant.
How many grams are in mass?
1,000 milligrams (mg) =1 gram10 centigrams =100 milligrams (mg)1 gram (g) =1,000 milligrams1,000 grams =1 kilogram (kg) = 1,000,000 mg1,000 kilograms =1,000,000 grams
How do you find the mass of an area?
- m= Total mass.
- A = Total area.
- ρA = Area density.
Is mass the same as weight?
Mass is a measure of the amount of material in an object, directly related to the number and type of atoms present in the object. … The unit of mass in the SI system is the kilogram (kg). Weight. In the trading of goods, weight is taken to mean the same as mass and is measured in kilograms.
How do you measure mass and weight?
Mass and weight are related by the equation Weight = mass x gravity. You might know this equation as Newton’s II Law of Motion Force = mass x acceleration (F = ma) where Force (or weight) is measure in Newtons, N, and the mass is measured in kilograms, kg.
How do you find mass with Newtons?
Plug the numbers you want to convert to mass into the new equation, m = F/a. As an example, we will use an object with a force of 10 N and an acceleration of 2 meters per second squared. Divide the newtons by the rate of acceleration, which will give you the mass of the object.
How do you find the mass number of molecules?
You look up the atomic mass of each of the elements in the formula, multiply it by the number of atoms of that element in the compound and add it to all the others. This gives you the mass, in grams, of one mole of the molecule.
What does 1 gram atom mean?
Hint: One gram atom means the mass of one mole of an element equal in grams to the atomic weight. One gram atom of oxygen is defined as the atoms present in one gram of oxygen atom.