What is the mechanism of sn1 and sn2

Nathan Sanders
Published Feb 16, 2026
Sn1 and Sn2 are the two forms of nucleophilic substitution reaction. SN1 involves one molecule while Sn2 involves two molecules. … There are two types of nucleophile: Neutral – Molecules that have a lone pair(s) of electrons but have an overall neutral charge are called neutral nucleophiles.
What is SN1 and SN2 reaction mechanism?
Sn1 and Sn2 are the two forms of nucleophilic substitution reaction. SN1 involves one molecule while Sn2 involves two molecules. … There are two types of nucleophile: Neutral – Molecules that have a lone pair(s) of electrons but have an overall neutral charge are called neutral nucleophiles.
What is SN1 reaction and its mechanism?
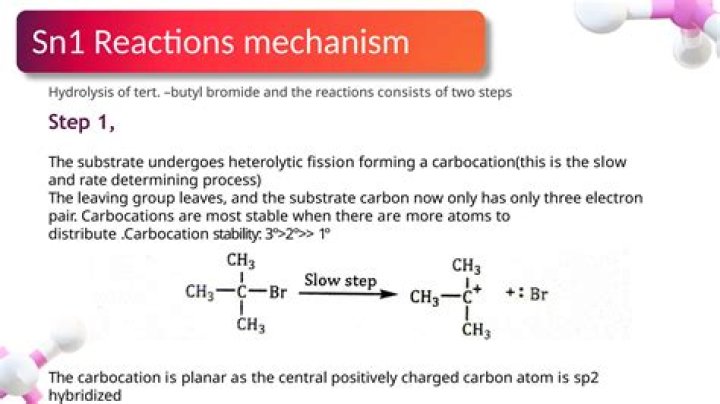
The SN1 reaction is a nucleophilic substitution reaction where the rate determining step is unimolecular. It is a type of organic substitution reaction. SN1 stands for substitution nucleophilic unimolecular. … The SN1 reaction is often referred to as the dissociative mechanism in inorganic chemistry.
What is the mechanism of SN2 reaction?
SN2 reaction mechanism requires the attack of nucleophile from the back side of the carbon atom. So the product assumes a stereochemical position opposite to the leaving group originally occupied. This is called inversion of configuration.What is SN2 mechanism explain with example?
The SN2 reaction – A Nucleophilic Substitution in which the Rate Determining Step involves 2 components. -SN2 reactions are bimolecular with simultaneous bond-making and bond-breaking steps. -SN2 reactions do not proceed via an intermediate. -SN2 reactions give inversion of stereochemistry at the reaction centre.
What is the order of kinetics in the SN1 mechanism?
It forms in the rate-determining step, which does not involve the nucleophile. In the second, fast step, the carbocation reacts with a nucleophile such as water to form the product. The rates of SN1 reactions decrease in the order tertiary > secondary > primary > > methyl.
Why is SN1 called SN1?
SN 1 reactions’ rates are only dependent on on entity, the electrophile (loss of a leaving group is the first step of this reaction and it does not require a nucleophile at first to have the Leaving Group leave and form carbocation), therefore it is called SN1.
Is SN1 a one step reaction?
No, an SN1 reaction involves two steps. In a typical nucleophilic substitution reaction, a nucleophile Nu⁻ attacks a substrate R-LG. The leaving group LG departs, and the Nu replaces (substitutes) it in the substrate.What is the mechanism of Wurtz reaction?
Mechanism One: By a formation of free radical as an intermediate. Step 2: In second step, second sodium atom release one more electron to the free radical and provide a carbonium ion. Step 3: A halide ion is displaced by alkyl anion from another molecule of alkyl halide. This reaction is considered as a SN2 reaction.
Which compound react most rapidly by SN1 mechanism?The correct answer is MeO – CH2 – Cl. MeO-CH2- Cl will react faster in an SN1 reaction with the OH- ion. This happens due to the stability of the carbocation in the compound.
Article first time published onDoes SN2 have inversion?
Purely SN2 reactions give 100% inversion of configuration. Thus SN2 reactions must occur through backside attack. The phrase “inversion of configuration” may lead you to believe that the absolute configuration must switch after SN2 attack.
What is the meaning of SN1?
SN1 Definition. The SN1 reaction – A Nucleophilic Substitution in which the Rate Determining Step involves 1 component. -SN1 reactions are unimolecular, proceeding through an intermediate carbocation. -SN1 reactions give racemization of stereochemistry at the reaction centre.
What is the full form of SN1 reaction?
From Wikipedia, the free encyclopedia. The SN1 reaction is a substitution reaction in organic chemistry, the name of which refers to the Hughes-Ingold symbol of the mechanism. “SN” stands for “nucleophilic substitution“, and the “1” says that the rate-determining step is unimolecular.
Which of the following alkyl halide is hydrolysed by SN1 mechanism?
CH3Cl.
Why is it called SN2?
In the SN2 reaction, the addition of the nucleophile and the departure of the leaving group occur in a concerted(taking place in a single step) manner, hence the name SN2: substitution, nucleophilic, bimolecular.
What affects SN2?
In order of decreasing importance, the factors impacting SN2 reaction pathways are. 1) structure of the alkyl halide. 2) strength of the nucleophile. 3) stability of the leaving group. 4) type of solvent.
What factors favor SN1 or SN2?
The SN2 Is Favored By Polar Aprotic Solvents. The SN1 Tends To Proceed In Polar Protic Solvents. … The SN1 reaction tends to proceed in polar protic solvents such as water, alcohols, and carboxylic acids, which stabilize the resulting (charged) carbocation that results from loss of the leaving group.
What is the order of kinetics in the SN2 mechanism?
The bimolecular nucleophilic substitution reaction follows second-order kinetics; that is, the rate of the reaction depends on the concentration of two first-order reactants.
What is the order of SN2 reaction?
In SN2 reaction,from experimental conclusions,it is observed that the rate of reaction depends upon concentration of substrate as well as concentration of nucleophile. Thus it is second order reaction.
Is SN2 first order?
The term SN2stands for Substitution reaction, Nucleophilic, 2nd order (also called bimolecular). … For this reason, the concentrations of both the nucleophile and the alkyl halide are proportional to the observed SN2 reaction rate.
Is Wurtz reaction free radical mechanism?
The Wurtz reaction occurs through a free-radical mechanism that makes possible side reactions producing alkene products.
What is Swarts?
Definition of swart 1a : swarthy. b archaic : producing a swarthy complexion. 2 : baneful, malignant.
Why is dry ether used in Wurtz reaction?
In Wurtz reaction sodium metal is used which is very reactive. So selection of the solvent is done in such a way that the sodium metal does not react with the solvent. Dry ether is a solvent which does not react with sodium. … Since dry ether is a good non-polar, aprotic solvent, so it is used in Wurtz reaction.
How many steps is SN2?
The SN2 Mechanism. The SN2 mechanism is a one-step process in which a nucleophile attacks the substrate, and a leaving group, L, departs simultaneously. Because the reaction occurs in one step, it is concerted. The substrate and the nucleophile are both present in the transition state for this step.
Why is sn1 faster than SN2?
SN2 take place faster . its a one steped process . and SN 1 is two steped process in which first step that is formation of carbcation is slow and second step that is attack of nucleophile is fast .
How many steps are there in an SN2 reaction?
Bimolecular nucleophilic substitution (SN 2) reactions are concerted, meaning they are a one step process. This means that the process whereby the nucleophile attacks and the leaving group leaves is simultaneous.
Which compound react most rapidly by SN2 mechanism?
In (CH3)3CCH2I (neopentyl iodide), in addition to iodine, the carbon atom is bound to two hydrogen atoms and only one other carbon. This is called a primary halide (primary meaning bound to only one carbon). You would therefore expect this compound to have the fastest SN2 reaction rate.
Which of the following compounds give SN2 mechanism?
CH3−Br.
Which halide reacts most rapidly via an SN2 mechanism?
3. The Reaction Rate Of The SN2 Reaction Is Fastest For Small Alkyl Halides (Methyl > Primary > Secondary >> Tertiary) Finally, note how changes in the substitution pattern of the alkyl halide results in dramatic changes in the rate of the reaction.
Does SN2 prefer primary or tertiary?
The “big barrier” to the SN2 reaction is steric hindrance. The rate of SN2 reactions goes primary > secondary > tertiary. The “big barrier” to the SN1 and E1 reactions is carbocation stability.
Can SN1 be primary?
Primary carbons can only be SN2 substitutions. Tertiary carbons can only be SN1. Secondary, benzyllic, or allylic carbons can be either SN1 or SN2. Here are examples of the types of carbons to look for.