What is the molecular geometry of SnCl2

Isabella Wilson
Published Feb 13, 2026
Formula# BondsMolecular geometryBF33Trigonal planarSnCl22BentCH44TetrahedralNH33Trigonal pyramidal
What is geometry of SnCl2?
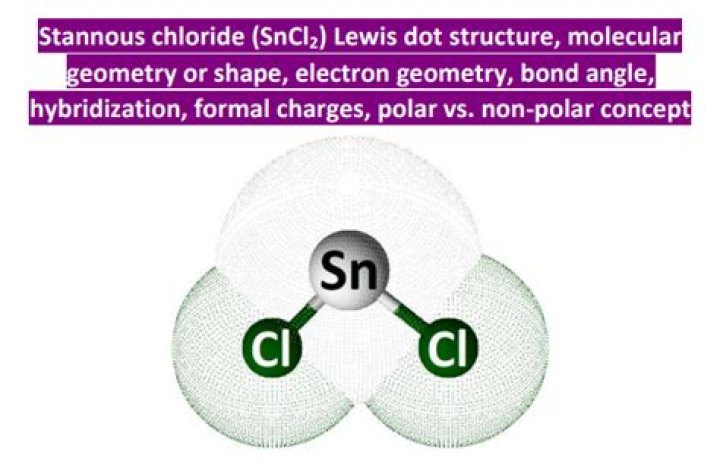
In the gaseous state, SnCl2 has sp2 hybridization. Therefore the geometry will be trigonal planar. but due to the presence of one lone pair, it forms a bent structure.
How many lone pairs are in SnCl2?
With 18 total valence electrons in the structure, and with 16 valence electrons being taken up by the two Chlorines with 3 lone pairs around each and 1 single bond linking each Cl to Sn thanks to the fact that Chlorine’s formal charge will be 0 when there are 6 valence electrons and 1 bond around it, 2 valence …
Is SnCl2 a linear?
SnCl2 is non – linear molecule.Is SnCl2 trigonal planar?
Out of these three hybrid orbitals, one contains a lone pair of electrons and two contain a bond pair of electrons. The electron pair geometry is trigonal planar and the molecular geometry is bent or angular.
Is SnCl2 polar or nonpolar?
On the other hand, SnCl2 is a bent molecule. Thus, its bond dipoles do not cancel and SnCl2 is a polar molecule.
Why is SnCl2 not linear?
In the case of Magnesium chloride(MgCl2), Mg is sp hybridized with a bond angle of 180o and hence is linear. whereas in the case of Stannous chloride(SnCl2), Sn is sp2 hybridized with a bond angle of 120o and hence the molecule is angular.
What is the electron geometry and molecular geometry for if4+?
The geometric shape of a tetrahedral molecule is created by its central atom bonding with four other atoms that surround it, creating angels of 190 degrees Fahrenheit.What is the molecular geometry of sf6?
The molecular geometry of SF6 is octahedral with a symmetric charge distribution around the central atom. Therefore this molecule is nonpolar. Sulfur Hexafluoride on Wikipedia.
What is the molecular geometry of PBr5?PBr5 ‘s molecular geometry is trigonal bipyramidal.
Article first time published onHow many bonds does SnCl2 have?
In SnCl2 two bonds are formed with one s and one p orbital and other p orbitals electrons remained unused.
What is the name of the compound SnCl2?
Tin(II) chloride, also known as stannous chloride, is a white crystalline solid with the formula SnCl2. It forms a stable dihydrate, but aqueous solutions tend to undergo hydrolysis, particularly if hot. SnCl2 is widely used as a reducing agent (in acid solution), and in electrolytic baths for tin-plating.
What is the bond angle in 50 SnCl2?
Both of these molecules have 3 regions of electron density: 2 bonding regions and one lone pair. As a result the geometry would be similar to that of a trigonal planar molecule but the angles would have to be <120 to account for the additional space taken up by the lone pair.
Is SnCl2 V shaped?
The shape of Sncl2 is V-shaped due to one lone pair according to VSPER theory..
Is SnCl2 ionic or molecular compound?
SnCl2 is ionic but SnCl4 is covalent.
Which of following molecule is not polar?
Any of the homonuclear diatomic elements: H2, N2, O2, Cl2 (These are truly nonpolar molecules.) Carbon dioxide – CO. Benzene – C6H. Carbon tetrachloride – CCl.
Is H3O+ polar or nonpolar?
The overall molecule is Polar because the shape of the molecule is Trigonal Pyramidal, which means it has the lone pair electrons. Becuase of the lone pair the pulling is unequal. H3O+ has 3 polar bonds. … So H3O+ has 3 polar bonds, and the overall molecule is polar too.
Why does sf6 have regular geometry?
SF6 molecular geometry will be octahedral because if we look at the structure sulphur hexafluoride has a central sulphur atom around which12 electrons or 6 electron pairs are present and there are no lone pairs. The F-S-F bonds are slated to be at 90 degrees.
What is the hybridization of P4?
P4 is white phosphorous in which each P is surrounded by 3 sigma bond and 1 lone pair , 4 orbitals require Therefore hybridisation of each phosphorous in P4 is SP3.
What is the hybridization for the C atom in cocl2?
${{Cl – (C = O) – Cl}}$ contains one double bond on a carbon atom. Hence, its hybridization is ${{s}}{{{p}}^{{2}}}$ .
How do you find the hybridization of SnCl2?
In $SnC{l_2}$, three atomic orbitals of tin take part in hybridization to create three degenerate hybrid orbits. The three atomic orbitals are one s-orbital and two p-orbital. Thus, the hybridization will be $s{p^2}$ and due to the presence of lone pairs the structure is bent and the resulting geometry is angular.
What is the pH of SnCl2?
SnCl2 has pH 1-2 (source 1, 2).
How do you make a SnCl2 solution?
Dissolve 5 g of the sample in 5 ml of hydrochloric acid, dilute to 50 ml with water, filter if not clear and heat the filtrate or clear solution to boiling. Add 5 ml of barium chloride TS, digest in a covered beaker on a steam bath for Page 2 2 h, and allow to stand overnight.
Why is SnCl2 a strong reducing agent?
-Sn doesn’t exhibit inert pair effect & thus higher oxidation state is more stable. … -During the reaction, SnCl2 gets oxidised while other compound is reduced. -Thus, SnCl2 is a good reducing agent.
In which of the following pairs bond angle is 109 28 '?
The bond angle in ammonia is 109∘28.