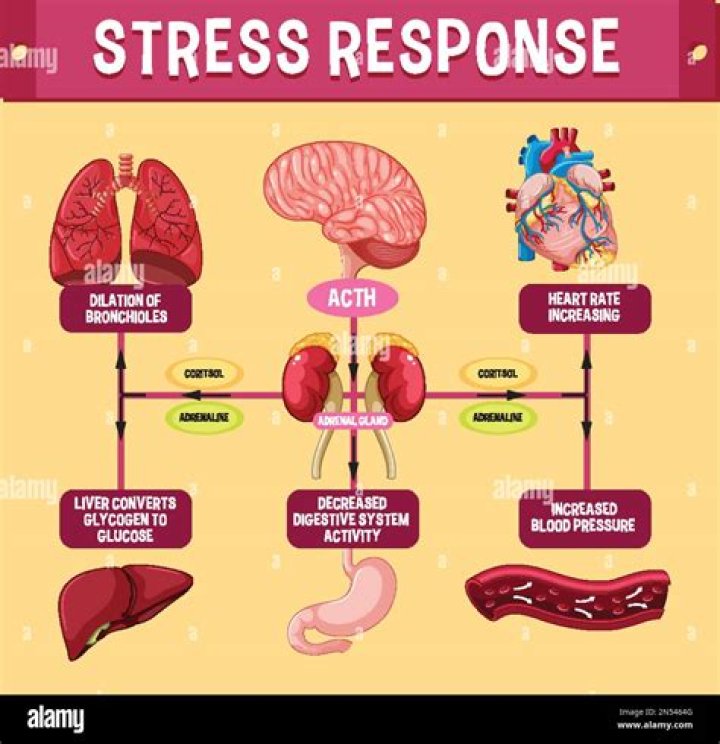
What is the number of neutrons in mercury

Nathan Sanders
Published Feb 24, 2026
One atom of Mercury has 121 neutrons. To find the number of neutrons in Mercury, you need to first find its atomic number on the Periodic Table of Elements
What is mercury number?
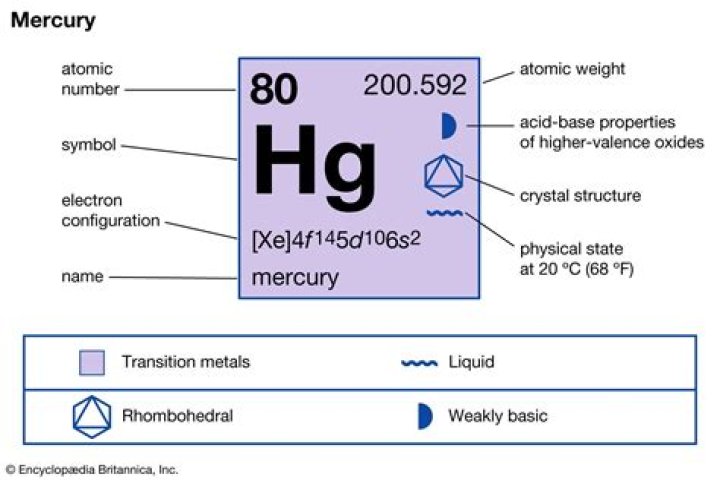
Mercury is a chemical element with symbol Hg and atomic number 80.
What is the neutron symbol?
ParticleSymbolChargeelectrone–1protonp++1neutronno0
How many neutrons are in Mercury 204?
Properties of Mercury-204 Isotope:MERCURY-204Neutron Number (N)124Atomic Number (Z)80Mass Number (A)204Nucleon Number (A)204How do u calculate atomic mass?
For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. This is because each proton and each neutron weigh one atomic mass unit (amu). By adding together the number of protons and neutrons and multiplying by 1 amu, you can calculate the mass of the atom.
Where do you find Mercury?
What is Mercury? Mercury is a naturally-occurring chemical element found in rock in the earth’s crust, including in deposits of coal. On the periodic table, it has the symbol “Hg” and its atomic number is 80.
What is Mercury formula?
Molecular Formula. Hg. Synonyms. 7439-97-6.
How many protons neutrons and electrons does Mercury 204 have?
Hg20480 has 80 protons 80 electrons and 124 neutrons. The mass of 204 is made up of the protons and neutrons.Why is Mercury called Mercury?
The Romans believed that gods and goddesses were in charge of everything on Earth. Mercury is named after the messenger for their gods. … The planet Mercury moves quickly around the sun. That is how it got its name.
How many neutrons are in CA?Johnson Z. In calcium-40, there are 20 neutrons.
Article first time published onHow many electrons neutrons and protons does Mercury have?
Mercury atoms have 80 electrons and 80 protons with 122 neutrons in the most abundant isotope.
What is the mass number of an isotope of Mercury that has 122 neutrons?
In the case of mercury, the most common isotope is mercury-202. Use the equation, 202-80=122, to find that mercury-202 has 122 neutrons.
What has 27 protons and 33 neutrons?
Cobalt-60 has 27 protons (thus 33 neutrons).
What is the mass of one neutron in amu?
mn = mass of a neutron (1.008665 amu)
What is the number of neutrons in Sulfur?
NameHydrogenNeutrons0Electrons1Atomic Number (Z)11
What are neutrons Class 9?
Neutrons are subatomic particles that are one of the primary constituents of atomic nuclei. They are usually denoted by the symbol n or no. Neutrons do not have any net electric charge associated with them. They do, however, have mass which is slightly greater in magnitude than that of a proton.
What is the number of neutrons in carbon?
Every carbon atom has six protons, and the majority of carbon atoms have six neutrons. A carbon-12 atom has 6 protons (6P) and 6 neutrons (6N). But some types of carbon have more than six neutrons. We call forms of elements that have a different number of neutrons, isotopes.
What is the size of a neutron?
A neutron also has about the same diameter as a proton, or 1.7×10−15 meters.
What does Amu stand for?
abbreviation. atomic mass unit (def.
What is the Iupac name for Mercury?
mercury (Hg), also called quicksilver, chemical element, liquid metal of Group 12 (IIb, or zinc group) of the periodic table.
What are the valencies of mercury?
hg has a valency of +1 and +2 in +1 valency it’s called mercurous and in +2 valency its called mercuric!
What happens if mercury touches gold?
Freddie Mercury may have had the golden voice, but real mercury, that endlessly entertaining and dangerous liquid metal, has the golden touch. That is, if it touches gold it will immediately break the lattice bonds of the precious metal and form an alloy in a process known as amalgamation.
Which country produce mercury?
Large commercial deposits of mercury have been mined at Almadén, Spain; Idrija, Slovenia; Monte Amiata, Italy; Santa Barbara, Peru; and New Almaden, California, U.S. The world’s leading producers of mercury are China, Kyrgyzstan, and Chile.
What is black Mercury?
The potent properties of mercury are stabilised by its trituration with ‘sulphur’ (gandhaka), which has a natural affinity to form a unique compound with mercury through a grinding process. This triturated black compound made of ‘purified’ mercury and sulphur is black mercury sulphide, called kajjali.
How big is Mercury vs Earth?
With a radius of 1,516 miles (2,440 kilometers), Mercury is a little more than 1/3 the width of Earth. If Earth were the size of a nickel, Mercury would be about as big as a blueberry. From an average distance of 36 million miles (58 million kilometers), Mercury is 0.4 astronomical units away from the Sun.
Who named planet Earth?
All of the planets, except for Earth, were named after Greek and Roman gods and godesses. The name Earth is an English/German name which simply means the ground. It comes from the Old English words ‘eor(th)e’ and ‘ertha’. In German it is ‘erde’.
How many neutrons are in Mercury 203?
So, there must be 201 – 80 = 121 neutrons. Since atoms are electrically neutral, there must be as many electrons as there are protons.
What group is Mercury in?
Group12−38.829°C, −37.892°F, 234.321 KBlockd13.5336Atomic number80200.592State at 20°CLiquid202HgElectron configuration[Xe] 4f145d106s27439-97-6
How many neutrons are in CA 41?
half-life (λ)100,000yProtons20Neutrons21
How many neutrons are found in calcium 37?
No. of neutrons = 40−20=20. Hence 20 neutrons are present in calcium.