What is the polarity of toluene

Ava Hall
Published Feb 26, 2026
SolventRelative Polaritytoluene0.099benzene0.111ether0.117methyl t-butyl ether (MTBE)0.124
What is the polarity of toluene? - Google Search
Nonpolar solvents include alkanes (pentane, hexane, and heptane) and aromatics (benzene, toluene, and xylene). Other common nonpolar solvents include acetic acid, chloroform, diethyl ether, ethyl acetate, methylene chloride, and pyridine.
Is toluene polar protic or aprotic?
N-Methyl-2-pyrrolidone (NMP), toluene, benzene, acetone, chloroform, and ether are some of the widely used aprotic products. Among these, toluene was the most extensively used product, followed by benzene.
Is toluene slightly polar?
Because it is non-symmetrical and composed of an aromatic group and an aliphatic group, toluene is very slightly polar, the ring being negative and the methyl group positive as shown by spectroscopy.How do you determine the polarity of a solvent?
One way you could actually calculate a solvent’s polarity is by using its dielectrict constant, which is used to measure how well the solvent can partly cancel the field strenght of the electric field of a particle added to it. The higher the value of a solvent’s dielectric constant, the more polar it will be.
Is toluene soluble in water?
It is also used as a solvent and antiknock additive for aviation gasoline. Pure toluene (melting point, -95° C [-139° F]; boiling point, 110.6° C [231.1° F]) is a colourless, flammable, toxic liquid, insoluble in water but soluble in all common organic solvents.
What is the polarity of isopropanol?
Most of isopropyl alcohol (the three carbons and the hydrogens attached to them) is non-polar.
Why is toluene more polar than benzene?
According to the table of the organic solvent polarity, the relative value for benzene is 0.111 and 0.099 for toluene. However ,theoretically speaking, benzene is non-polar due to its highly structural symmetry, and toluene is polar because of methyl group’s electron donating character.Is toluene polar than hexane?
toluene has an sp3 hybridized carbon attached to an sp2 carbon. since this sp2 carbon has more s character than the sp3 carbon (33% to sp3’s 25%), it comparatively more electronegative and can actually withdraw electron density from the sp3 carbon.
Is Ligroin polar or nonpolar?Since ligroin is a nonpolar solvent (composed of 6-carbon alkanes), nonpolar sample molecules will easily dissolve in the solvent and not adsorb to the polar silica gel.
Article first time published onWhat type of molecule is toluene?
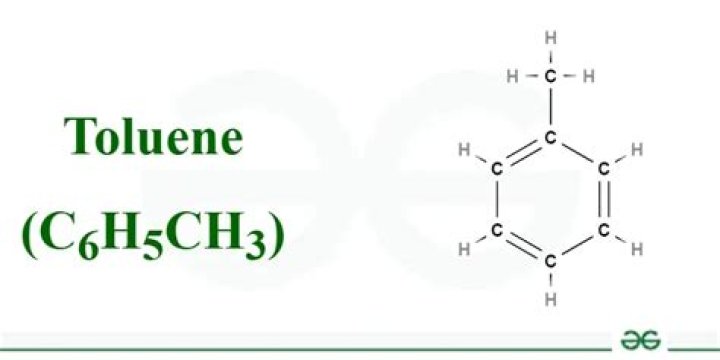
The Toluene Molecule. Toluene also known as toluol, is an aromatic hydrocarbon. It is a colorless, water-insoluble liquid with the smell associated with paint thinners. It is a mono-substituted benzene derivative, consisting of a CH3 group attached to a phenyl group.
Which solvent is non-polar?
Non-polar solvents cannot dissolve polar compounds since no opposite charges exist and the polar compound is not attracted. It is this absence of partial charge that also makes these molecules “non-polar”. Some of the examples of non-polar solvents include hexane, pentane, toluene, benzene, etc.
Is heptane a polar molecule?
Heptane (and its many isomers) is widely used in laboratories as a non-polar solvent.
How do you determine the polarity of a solution?
Solution Method Mixing a liquid of unknown polarity with water can tell you if the molecules in the liquid are polar or non-polar. Simply mix the liquid with an equal part of water and allow the mixture to sit undisturbed. Examine the mixture after the liquids have sat together for a time.
What is the polarity index?
Polarity Index (p1) Defined as a measure of the ability of the solvent. to interact with various test solutes.
What is the most polar solvent?
SolventRelative Polarityethylene glycol0.79trifluoroethanol0.898hexafluoroisopropanol0.969water1
What is the polarity of dichloromethane?
SolventPolarity IndexDipole momentDichloromethane3.11.141,2-Dichloroethane3.51.83Isopropyl Alcohol3.91.66n-Butyl Acetate4.01.84
What is the polarity of chloroform?
Polarity Index (P´)Methyl Isoamyl Ketone4.0n-Propyl Alcohol4.0Tetrahydrofuran4.0Chloroform4.1
Why is ethyl acetate polar?
Polar molecule contains bond dipoles, which do not cancel each other. Therefore, CH3COOCH2CH3 contains two polar bonds(CO and CO) in which their bond dipoles do not cancel each other. Hence, ethyl acetate is a polar compound. NB: Relative electronegativity determines whether a compound is a polar or not.
Is toluene hydrophobic or hydrophilic?
HPLC Separation of 8 Generic Compounds on Primesep Columns In this application, neutral hydrophilic (uracil, phenol and hydroquinone), neutral hydrophobic (toluene), hydrophilic acidic (benzoic acid), hydrophilic basic (lutidine) and hydrophobic basic (amitriptyline) are separated using gradient of ACN.
How soluble is toluene?
Toluene is a colorless, flammable liquid that is about 15% lighter than water. … At 25 °C, toluene has a solubility in water of 526 mg l−1, an estimated vapor pressure of 28.4 mm Hg and a Henry’s Law Constant of 6.64 × 10−3 atm-m3 mol−1(USEPA, 2011). The log octanol/water partition coefficient (Kow) is 2.73.
Why is toluene insoluble?
It is an aromatic hydrocarbon because there is no functional group in toluene. The molecular mass of toluene is 92.141 g /mol. Since it is an aromatic compound, its odor is sweet and benzene-like. … Hence, toluene is not soluble in water.
What is the polarity of a water molecule?
Polarity: Although the net charge of a water molecule is zero, water is polar because of its shape. The hydrogen ends of the molecule are positive and the oxygen end is negative. This causes water molecules to attract each other and other polar molecules.
What is relative polarity?
The relative polarity of a molecule may be represented by the values of the intrinsic states of the atoms. The same approach can yield a numerical value for a fragment or group within the molecule. One approach is to derive a numerical value that is relative to a very low and a very high polarity value.
Are hexanes polar?
Hexane is a non-polar solvent with a boiling point of 68°C, and is therefore the solvent of choice for oil extraction from rice bran to yield rice bran oil (RBO).
Are phenols polar?
Since Oxygen is more electronegative so it’s attract the electrons of O-H bond and hence there is some polarity and hence phenol is polar.
What is the polarity of octane?
Polarity IndexSolventBP (°C)0.4i-octane99.20.4octane99.21.7butyl ether142.21.7carbon tetrachloride76.5
Is ethyl acetate polar or nonpolar?
The ethyl acetate molecule, with the simplified formula C4 H8 O2 , is a polar solvent.
Is acetylsalicylic acid polar?
Acetylsalicylic acid (ASA) has two polar regions, a carboxyl group (giving it it’s acidic property) and a ester group. However, these two groups are attached to a benzene ring which is a non-polar 6 carbon hydrocarbon.
What is Ligroin in organic chemistry?
Ligroin is the petroleum fraction consisting mostly of C7 and C8 hydrocarbons and boiling in the range 90‒140 °C (194–284 °F). The fraction is also called heavy naphtha. Ligroin is used as a laboratory solvent. Products under the name ligroin can have boiling ranges as low as 60‒80 °C and may be called light naphtha.
Is water polar?
Water is a Polar Covalent Molecule The unequal sharing of electrons between the atoms and the unsymmetrical shape of the molecule means that a water molecule has two poles – a positive charge on the hydrogen pole (side) and a negative charge on the oxygen pole (side).