What is the process of nitrogen cycle

Lily Fisher
Published Apr 07, 2026
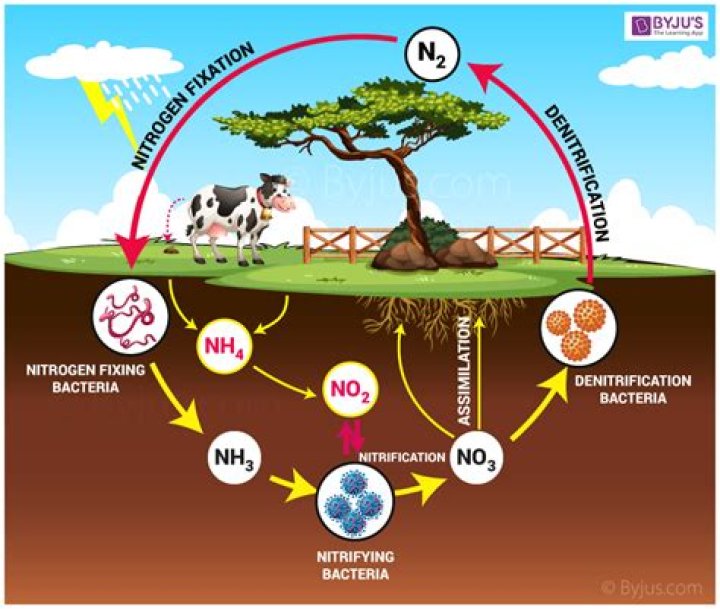
Nitrogen Cycle is a biogeochemical process through which nitrogen is converted into many forms, consecutively passing from the atmosphere to the soil to organism and back into the atmosphere. It involves several processes such as nitrogen fixation, nitrification, denitrification, decay and putrefaction.
What are the 4 stages of the nitrogen cycle?
- Nitrogen Fixation.
- Ammonification/ Decay.
- Nitrification.
- De-nitrification.
What is nitrogen cycle in simple words?
Definition of nitrogen cycle : a continuous series of natural processes by which nitrogen passes successively from air to soil to organisms and back to air or soil involving principally nitrogen fixation, nitrification, decay, and denitrification.
What are the 3 steps of the nitrogen cycle and what happens at each step in the process?
Overview: The nitrogen cycle involves three major steps: nitrogen fixation, nitrification, and denitrification. It is a cycle within the biosphere which involves the atmosphere, hydrosphere, and lithosphere. Nitrogen is found in several locations, or reservoirs.What are the 5 steps of the nitrogen cycle?
There are five stages in the nitrogen cycle, and we will now discuss each of them in turn: fixation or volatilization, mineralization, nitrification, immobilization, and denitrification.
What is the last step in the nitrogen cycle?
Step 4- Denitrification– Denitrification is the final step in which other bacteria convert the simple nitrogen compounds back into nitrogen gas (N2 ), which is then released back into the atmosphere to begin the cycle again.
How many steps does the nitrogen cycle have?
In general, the nitrogen cycle has five steps: Nitrification (NH3 to NO3-) Assimilation (Incorporation of NH3 and NO3- into biological tissues) Ammonification (organic nitrogen compounds to NH3) Denitrification(NO3- to N2)
Why is it called nitrogen cycle?
The nitrogen cycle is the biogeochemical cycle describing how nitrogen moves through the biosphere and atmosphere. … When living organisms die, nitrogen returns to the soil through ammonification. Nitrification by bacteria transforms ammonia in the soil into nitrates so that it is available for plants to use again.How does the nitrogen cycle work simple?
Nitrogen Cycle is a biogeochemical process through which nitrogen is converted into many forms, consecutively passing from the atmosphere to the soil to organism and back into the atmosphere. It involves several processes such as nitrogen fixation, nitrification, denitrification, decay and putrefaction.
Which of the following process explain how plants get nitrogen?Plants get their nitrogen from the soil and not directly from the air. … The act of breaking apart the two atoms in a nitrogen molecule is called “nitrogen fixation“. Plants get the nitrogen that they need from the soil, where it has already been fixed by bacteria and archaea.
Article first time published onWho discovered nitrogen cycle?
Nitrogen was officially discovered in 1772 by Scottish scientist Daniel Rutherford [6]. At the same time however, Carl Scheele, Henry Cavendish, Joseph Priestley and others were investigating ‘burnt or dephlogisticated air’, as air without oxygen was then called.
What is the role of decomposers in the nitrogen cycle?
The decomposers, certain soil bacteria and fungi, break down proteins in dead organisms and animal wastes, releasing ammonium ions which can be converted to other nitrogen compounds. … Nitrates are reduced to nitrogen gas, returning nitrogen to the air and completing the cycle.
How is nitrogen formed?
On a small scale, pure nitrogen is made by heating barium azide, Ba(N3)2. Various laboratory reactions that yield nitrogen include heating ammonium nitrite (NH4NO2) solutions, oxidation of ammonia by bromine water, and oxidation of ammonia by hot cupric oxide.
What Colour is nitrogen?
ElementColor NameRGB ValuesNitrogenlight blue[143,143,255]Sulfuryellow[255,200,50]Phosphorusorange[255,165,0]Chlorinegreen[0,255,0]
What is the role of plants in nitrogen cycle?
Plants absorb nitrates from the soil to make proteins. Animals consume plants and use it to form animal protein. Humans contribute to the cycle by adding nitrogen rich fertilisers to the soil and by using manure (The Physics Teacher, 2018).
Which processes remove nitrogen from the atmosphere?
Denitrification. Denitrification is the process that converts nitrate to nitrogen gas, thus removing bioavailable nitrogen and returning it to the atmosphere.
Which is the possible path of movement of nitrogen in the nitrogen cycle?
Five main processes cycle nitrogen through the biosphere, atmosphere, and geosphere: nitrogen fixation, nitrogen uptake through organismal growth, nitrogen mineralization through decay, nitrification, and denitrification.
How is nitrogen formed in atmosphere?
Nitrogen fixation is carried out by bacteria, algae and human activity, and once organisms have benefited from it, some of the nitrogen compounds break down and go back into the atmosphere as nitrogen gas.
What is nitrogen made up of?
Nitrogen is an element that can combine with itself or with other elements to make different compounds. For instance nitrogen gas, N2, is a compound made when two nitrogen atoms form a chemical bond. It makes up about 80% of the atmosphere, while oxygen gas, O2, makes up a little less than 20% of the atmosphere.
What period is nitrogen in?
Group15Melting pointPeriod2Boiling pointBlockpDensity (g cm−3)Atomic number7Relative atomic massState at 20°CGasKey isotopes
What Colour is water?
The water is in fact not colorless; even pure water is not colorless, but has a slight blue tint to it, best seen when looking through a long column of water. The blueness in water is not caused by the scattering of light, which is responsible for the sky being blue.
What Luster is nitrogen?
Nitrogen is a nonmetal. Like other elements in this group, it is a poor conductor of heat and electricity and lacks metallic luster in solid form.
Who discovered liquid nitrogen?
Nitrogen was first liquefied at the Jagiellonian University on 15 April 1883 by Polish physicists Zygmunt Wróblewski and Karol Olszewski.



