What is the product of zinc and sulfur

Rachel Hunter
Published Mar 12, 2026
They are mixed (carefully) and the mixture is stable sitting of the laboratory bench. When the mixture is touched with a heated metal rod, however, a violent reaction occurs (the reaction is termed exothermic; producing heat) and zinc sulfide is formed as the product.
What is zinc and sulfur called?
Zinc sulfide (or zinc sulphide) is an inorganic compound with the chemical formula of ZnS. This is the main form of zinc found in nature, where it mainly occurs as the mineral sphalerite.
What product's would you expect to be formed from the reaction between zinc and sulfur?
Zinc metal reacts with yellow crystals of sulfur in a fiery reaction to produce a white powder of zinc sulfide.
What is the formula for zinc and sulfur?
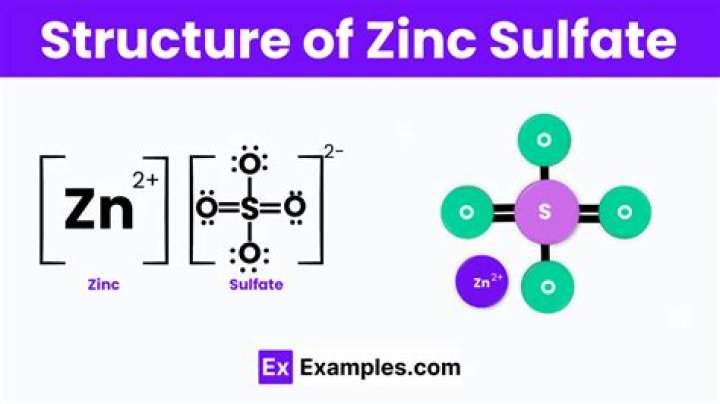
Formula and structure: The chemical formula of zinc sulfide is ZnS, and its molar mass is 97.47 g/mol. The chemical structure of ZnS is simple and consists of the zinc metal attached to a sulfur atom through a polar covalent bond.What is the name for ZnS?
CAS Reg. No.1314-98-3SciFinder nomenclatureZinc sulfide (ZnS)Empirical formulaSZnMolar mass97.47 g/molAppearanceWhite, grayish-white, or yellow crystals or powder
Is ZnS FCC or BCC?
Zincblende/sphalerite is based on a fcc lattice of anions whereas wurtzite is derived from an hcp array of anions. In both structures, the cations occupy one of the two types of tetrahedral holes present.
What is the formula of zinc sulphate?
Zinc sulfate is the inorganic compound with the formula ZnSO4 and historically known as “white vitriol”.
What are the products of the reaction of the Zn s →?
Zn (s) + S (s) → ZnS (s) When the mixture is touched with a heated metal rod, however, a violent reaction occurs (the reaction is termed exothermic; producing heat) and zinc sulfide is formed as the product.What solid does ZnS form?
SiteZnSCorner08(1/8) = 1Total44
What are the products in the following chemical equation Zn cuso4?1. Zinc and copper (II) sulphate. The substances that undergo chemical change in the reaction are the reactants. The new substance formed during the reaction is the product.
Article first time published onWhat is the color of the product of the reaction of zinc and sulfur?
Zinc (Zn) is a silver-gray element that can be ground into a powder. If zinc is mixed at room temperature with powdered sulfur (S), a bright yellow element, the result will simply be a mixture of zinc and sulfur. No chemical reaction occurs.
Does zinc contain sulfur?
Zinc sulfate is a combination of sulfur and zinc. … Zinc is an essential mineral for human, animal, and plant nutrition. Zinc can be found naturally in the environment, foods, and water. Products that contain zinc have been registered for use in pesticide products in the United States since 1973.
How is zinc oxide made from zinc sulfide?
At temperatures between about 230–430 °C (446–806 °F), H2S is converted to water by the following reaction: H2S + ZnO → H2O + ZnS. The zinc sulfide (ZnS) is replaced with fresh zinc oxide when the zinc oxide has been consumed.
What does oxygen sulfur make?
Sulfur reacts with oxygen to form sulfur dioxide. When nonmetals react with one another, the product is a molecular compound.
What is the chemical name of zinc blade?
PubChem CID14821StructureFind Similar StructuresChemical SafetyLaboratory Chemical Safety Summary (LCSS) DatasheetMolecular FormulaSZn or ZnSSynonymsZINC SULFIDE 1314-98-3 Sachtolith Sphalerite sulfanylidenezinc More…
Is ZnS fluorescence?
Pure ZnS doesn’t fluoresce. The stuff used as a phosphor has a little cadmium and silver added to it. These impurities are stuck in the latice and the electrons associated with them have a different set of permitted energy levels from the Zn atoms.
What products contain zinc sulfate?
- Livestock Feed. Zinc sulfate monohydrate is a readily-accessible form of zinc supplementation and is commonly used as an additive for livestock feed. …
- Multivitamins. …
- Rehydration Kits For Oral Rehydration Therapy (ORT) …
- Roof Cleaning Soaps & Sprays. …
- Toothpaste.
What is zinc monohydrate?
Zinc sulfate monohydrate is an inorganic compound that was historically known as “white vitriol,” and it has a variety of uses in medicine, industry, and more. In this guide from Bisley, we’ll take a look at a few common uses for this colourless, crystalline compound, and explain how it’s used.
What is the name for AuCl3?
Auric trichloride. Gold chloride (AuCl3) Gold(III)chloride.
How many units is ZnS?
If half of the tetrahedral holes in the ccp structure of sulfide ions were filled with zinc ions, one would obtain the ZnS structure. Both, zinc and sulfur have tetrahedral coordination. There are four formula units of ZnS per unit cell (S: 8 corners (8/8) + 6 faces (6/2), Zn: 4 tetrahedral holes (4/1)).
How do you make zinc sulphide?
Zinc sulfide is often produced by reacting zinc oxide with hydrogen sulfide, which produces zinc sulfide and water. Another method for producing the chemical is by heating a solution of zinc sulfate with sodium thiosulfate. A third way is to make a equimolar mix of zinc and sulfur powders and ignite the mix.
Is zinc sulphide a phosphorescent?
Zinc sulfide, with addition of few ppm of suitable activator, exhibits strong phosphorescence, and is currently used in many applications, from cathode ray tubes through X-ray screens to glow in the dark products.
Is zinc sulfide A FCC?
ZnS, Zinc Sulphide, Zinc Blend, Wurtzite In the zinc blend structure the sulphur ions form an fcc structure and the zinc ions occupy half of the tetrahedral sites in this structure to attain charge neutrality. The crystal has a lattice parameter of 0.541 nm.
Is ZnS ionic or covalent?
ZnS (zinc sulfide) is an ionic compound. This is because ionic compounds are made of metals and nonmetals.
What is zinc blende formula?
Due to the same number of atoms in a single unit cell of Zn and S, it is consistent with the formula ZnS. So, this will be the molecular or chemical formula of Zinc Blende. Hence, the formula of Zinc Blende is ZnS.
Is ZnS an ionic solid?
ZnS is a / an ionic ionic crystal.
What is zinc Sulphuric acid?
Zinc reacts with dilute sulphuric acid to form zinc sulphate and hydrogen gas is evolved. Since the products zinc sulphate and hydrogen gas are entirely different in chemical composition and chemical properties from the reactants, the reaction is a chemical change.
What is zinc powder?
Zinc powder is a bluish-gray colored, pure metal powder. It is produced when purified vapors of zinc are condensed. … Zinc powder is utilized for many different purposes in various fields.
What are the products of zinc and lead nitrate?
1) Zinc and aqueous lead (II) nitrate react to form aqueous zinc nitrate and lead metal. nitrate + lead metal. 2) Aqueous aluminum bromide and chlorine gas react to form aqueous aluminum chloride and bromine gas.
What are the products in the equation?
Products are the end-result of the reaction and are written on the right-hand side of the equation.
What type of reaction is CuSO4 and Zn?
CuSO4(aq) + Zn(s)→ ZnSO4(aq) + Cu(s) is an example of decomposition reaction.