What is the purpose of the Institutional Review Board IRB quizlet

Rachel Hunter
Published Apr 16, 2026
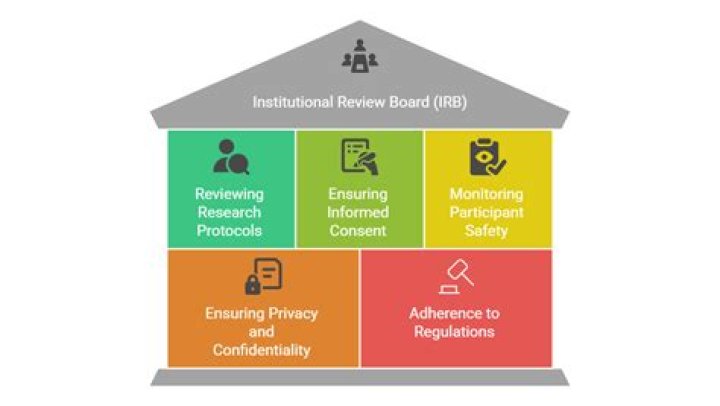
Institutional review boards (IRBs) are tasked with reviewing all studies involving human subjects to protect their rights and welfare.
What is the purpose of the Institutional Review Board IRB?
This group review serves an important role in the protection of the rights and welfare of human research subjects. The purpose of IRB review is to assure, both in advance and by periodic review, that appropriate steps are taken to protect the rights and welfare of humans participating as subjects in the research.
What are institutional review boards and what is the purpose of one quizlet?
An IRB consists of a group of scholars from various departments who are mandated to review proposals to ensure that they comply with ethical guidelines and write a summary of proposed risks to subjects. You just studied 2 terms!
What is the Institutional Review Board IRB quizlet?
Institutional review board (IRB) definition: … The Institutional Review Board (IRB) means any board, committee, or other group formally designated by an institution to review, to approve the initiation of, and to conduct periodic review of, biomedical research involving human subjects.Which of the following best describes the purpose of institutional review board?
The purpose of IRB review is to assure, both in advance and by periodic review, that appropriate steps are taken to protect the rights and welfare of humans participating as subjects in the research.
What are the responsibilities of an IRB IEC?
An IRB/IEC reviews the appropriateness of the clinical trial protocol as well as the risks and benefits to study participants. It ensures that clinical trial participants are exposed to minimal risk in relation to any benefits that might result from the research.
Why was the institutional review board created?
On July 12, 1974, the National Research Act established the existence of IRBs to review biomedical and behavioral research involving human subjects. In March 1983, federal regulations detailing the basic U.S. Department of Health and Human Services policy for the protection of human research subjects were adopted.
What is the frequency of the IRB IEC review required according to ICH GCP?
The IRB/IEC should conduct continuing review of each ongoing trial at intervals appropriate to the degree of risk to human subjects, but at least once a year.What is the purpose of a research review committee quizlet?
-a committee that has been formally designated to approve, monitor, and review biomedical and behavioral research involving humans with the aim to protect the rights and welfare of the research subjects.
What is an institutional review board in psychology?Institutional Review Boards (IRBs) are federally-mandated, locally-administered groups charged with evaluating risks and benefits of human participant research at their institution. … Federal law and APA standards require IRB review of all human participant research projects.
Article first time published onWho sits on an institutional review board?
An Institutional Review Board (IRB) is a committee made up of individuals who have training in scientific areas, individuals who have expertise and training in non-scientific areas, and members of the community who may represent people who would participate as subjects in research studies.
What information should be provided to an IRB for review at the initiation of a study?
The study protocol (and amendments), the information to be given to the subject (informed consent, advertisements), the Investigator Brochure (or drug label), any other relevant safety information, and an outline of the qualifications of the investigator.
Which of the following by law is a required characteristic of institutional Review Board IRB membership composition?
An IRB consists of at least five members of varying backgrounds. IRB members should have the professional experience to provide appropriate scientific and ethical review. An IRB must have at least one scientist member and at least one member whose primary concerns are nonscientific.
Which statement best describes the role of IRB?
Terms in this set (5) Which statement best describes the role of an IRB: a committee that reviews different types of human subjects research.
What does the IRB do to protect a research participant from harm?
The IRB is a primary mechanism for institutional protection of human subjects. … Under both DHHS and FDA regulations, the IRB has responsibility to approve or disapprove all covered research activity, requiring for instance that subjects are given enough information to be able to provide informed consent.
How does the IRB protect human subjects?
At UNH, the primary purpose of the Institutional Review Board for the Protection of Human Subjects in Research (IRB) is to protect the rights and welfare of human research subjects by ensuring that physical, psychological, legal, and/or social risks to subjects are minimized, and when present, justified by the …
What is the composition of Institutional Review Board IRB?
It is recommended that the IRB/IEC should include: (a) At least five members. (b) At least one member whose primary area of interest is in a nonscientific area. (c) At least one member who is independent of the institution/trial site.
What is the purpose of investigator's brochure?
The purpose of the IB is to compile data relevant to studies of the IP in human subjects gathered during preclinical and other clinical trials. An IB is intended to provide the investigator with insights necessary for management of study conduct and study subjects throughout a clinical trial.
What is the purpose of a research review committee?
The purpose of the Research Review Committee (RRC) is to enhance the scientific quality of research proposals. The major function of the Research Review Committee (RRC) is to ensure that research proposals prepared by investigators meet the highest scientific and ethical standards.
Is Institutional Review Board capitalized?
Many simply capitalize the term “Institutional Review Board” as the proper name of their instance. Regardless of the name chosen, the IRB is subject to the FDA’s IRB regulations when studies of FDA-regulated products are reviewed and approved.
What does informed consent do quizlet?
True:informed consent means a decision made by or for a patient after being informed about the proposed procedures, risks, benefits, and alternatives to the proposed treatment. … The ability of the patient to understand the risks, benefits, and alternatives to the procedure and make a decision based on that information.
What must the IRB IEC review and approval or grant favorable opinion before trial start?
Good Clinical Practice: Definition and Historical Background Since clinical studies involve the participation of human beings, compliance with the GCP standards is crucial to ensure the safety, well-being, and confidentiality of all trial subjects.
Which of the following documents should an IRB IEC review to satisfy this responsibility?
The IRB/IEC should obtain the following documents: trial protocol(s)/amendment(s), written informed consent form(s) and consent form updates that the investigator proposes for use in the trial, subject recruitment procedures (e.g. advertisements), written information to be provided to subjects, Investigator’s Brochure …
What is an institutional review board quizlet psychology?
IRB. a committee that reviews proposals for research with human participants in order to ensure that the project conforms to accepted ethical standards.
Which institutional criteria are used to review an IRB application?
Criteria for IRB Approval of Research Risks to subjects are reasonable in relation to the anticipated benefits … Informed consent will be sought from each prospective subject or the subject’s legally authorized representative … as required by #46.116.
What is the main focus of NIH's conflict of interest policy?
A conflict of interest policy is used to outline procedures for employees when a possible conflict exists between their own personal interests and the interests of the organization. The main focus of NIH’s conflict of interest policy is basically to prevent financial conflicts of interest.
What are the three principles discussed in the Belmont Report?
Three basic principles, among those generally accepted in our cultural tradition, are particularly relevant to the ethics of research involving human subjects: the principles of respect of persons, beneficence and justice.
What is the primary responsibility of oversight bodies such as an IRB or Iacuc )?
The primary responsibility of oversight bodies (such as an IRB or IACUC) is to assess legal-regulatory compliance, and if applicable, to protect research subjects.



