What is tonicity in biology quizlet

Ava Hall
Published Mar 27, 2026
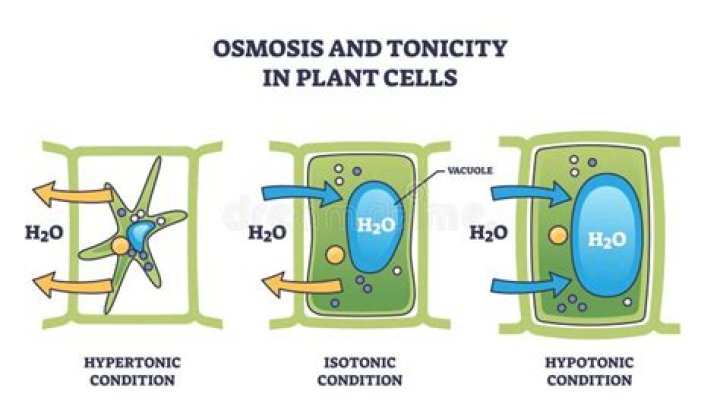
Tonicity. A description of the relative solute concentration in a solution as compared to another solution. Equilibrium. The condition that exists in system when there is a relatively equal distribution of a particular molecule. Hypertonic.
What is tonicity in biology?
Tonicity is defined as the ability of a solution surrounding a cell to cause that cell to gain or lose water (Urry et al., 2017). While osmolarity is an absolute quantity, tonicity is relative. … If two solutions have the same concentration of nonpenetrating solutes, they are considered isotonic.
What is tonicity example?
EXAMPLES. Tonicity is the reason why salt water fish cannot live in fresh water and vice versa. … If you place a salt water fish in fresh water, which has a low osmolarity, water in the environment will flow into the cells of the fish, eventually causing them to burst and killing the fish.
What is tonicity in simple terms?
Tonicity is the concentration of a solution as compared to another solution. Concentration describes the amount of solutes dissolved by a solution. … In biology, the tonicity of the environment compared to the cell determines how water moves across the semipermeable membrane.What is the difference between tonicity and osmolarity quizlet?
Osmolarity can be used to compare any 2 solutions. Tonicity always compares a solution and a cell, and tonicity is used to describe only the solution.
What is tonicity in plants?
Tonicity is a measure of the relative concentration of solute particles on either side of a semi-permeable membrane (e.g. inside a cell versus outside the cell). The higher the tonicity the greater the difference in the concentration of solutes (dissolved substances) and therefore the concentration of water. …
What is tonicity in biochemistry?
Tonicity. Tonicity is the capability of a solution to modify the volume of cells by altering their water content. The movement of water into a cell can lead to hypotonicity or hypertonicity when water moves out of the cell.
What is tonicity in biology class 9?
Tonicity is the relative concentration of the solutions that determine the direction and the extent of diffusion. … Isotonic Solution: The relative concentration of water molecules and the solute on either side of the cell membrane is the same.What is tonicity of a cell?
Tonicity. Tonicity is the capability of a solution to modify the volume of cells by altering their water content. The movement of water into a cell can lead to hypotonicity or hypertonicity when water moves out of the cell.
How does tonicity affect the function of the cell?The tonicity of a solution is related to its effect on the volume of a cell. A hypotonic solution causes a cell to swell, whereas a hypertonic solution causes a cell to shrink. … Although it is related to osmolality, tonicity also takes into consideration the ability of the solute to cross the cell membrane.
Article first time published onWhat is tonicity in physiology?
The ability of an extracellular solution to make water move into or out of a cell by osmosis is known as its tonicity. Tonicity is a bit different from osmolarity because it takes into account both relative solute concentrations and the cell membrane’s permeability to those solutes.
What is tonicity and osmosis?
“Tonicity is the ability of a solution to affect the fluid volume and pressure in a cell. If a solute cannot pass through a plasma membrane, but remains more concentrated on one side of the membrane than on the other, it causes osmosis.”
What is tonicity in muscle?
the state or quality of having good muscular tone or tension. — tonic, adj. See also: Health. the state or quality of having good muscular tone or tension.
What's the difference between osmolarity and tonicity?
Osmolarity and tonicity are related but distinct concepts. … The terms are different because osmolarity takes into account the total concentration of penetrating solutes and non-penetrating solutes, whereas tonicity takes into account the total concentration of non-freely penetrating solutes only.
What factors are responsible for the tonicity of a solution as compared to a cell?
What factors are responsible for the tonicity of a solution as compared to a cell? The osmotic pressure of a solution is usually a factor in tonicity. A cell is placed in a 15% solute solution and the cell shrinks.
Which of the following factors affect the tonicity of a solution surrounding a cell?
What factors are responsible for the tonicity of a solution? Number and type of solutes present and membrane permeability.
What is tonicity in pharmacy?
Tonicity is a property of a solution in reference to a particular membrane, and is equal to the sum of the concentrations of the solutes which have the capacity to exert an osmotic force across the membrane.
How is tonicity measured?
Tonicity is a measure of the relative concentration of non-penetrating solute on either side of a membrane. … Molarity is equal to the number of moles of solute divided by the number of liters of solvent, so divide the number of moles by the number of liters of solution to find the molarity.
How does tonicity impact nutrient intake in cells?
Tonicity refers to the concentration of carbohydrates (sugars) and electrolytes in a solution compared to the concentration of these components in human blood. This, in turn, affects how quickly the body can absorb its fluids and nutrients, impacting hydration and energy levels differently depending on tonicity.
What is the tonicity of the solution that the plant cells is in?
Plant cells have a cell wall around the outside than stops them from bursting, so a plant cell will swell up in a hypotonic solution, but will not burst.
What is the medical importance of tonicity?
Having a good understanding of tonicity gives a good insight into how dehydration can affect the cells of the body. An isotonic solution is best because the osmotic pressure within and outside the cell is equal, so there is no net movement of water and the cells will retain their shape and function.
How does tonicity affect animal cells?
The effects of Tonicity by means of different solutions are given below: Effect on living animal cells: The animal cells, such as red blood cells, will become crenated. When placed in a hypertonic solution, their appearance will consist of an irregular surface (water moves external to the cell).
What cells need this tonicity to be at homeostasis?
Animal cells need to be in isotonic solutions to be at homeostasis. Homeostasis is a balance and is important for all living things to maintain. …
Does glucose contribute to tonicity?
Tonicity depends only on the concentration of nonpenetrating solutes, so any solution of pure glucose will be hypotonic, no matter what its osmolarity, and tonicity describes only the change in cell volume at equilibrium.
What are vacuoles Class 9?
They are storage sacs for solid or liquid content.In animals cell,they are small and temporary. They store water,glycogen and proteins,single membrane maintain water balance.
What is Endosmosis?
Endosmosis is the movement of the water inside the cells when the cell is placed in a hypotonic solution. This movement of water causes the cell to swell.
How does tonicity affect red blood cells?
The tonicity of the urea and resultant osmotic water movement results in hemolysis of the cells, and this will form the blood used for the production of the hemoglobin standards that will be used to assess the degree of hemolysis in the experiment.
What fluids have the same tonicity as plasma?
Isotonic IV Fluids Most IV fluids are isotonic, meaning, they have the same concentration of solutes as blood plasma. When infused, isotonic solutions expand both the intracellular fluid and extracellular fluid spaces, equally.
How is tonicity related to osmolarity?
Tonicity is equal to the osmolality less the concentration of these ineffective solutes and provides the correct value to use. Osmolality is a property of a particular solution and is independent of any membrane. Tonicity is a property of a solution in reference to a particular membrane.
What is the tonicity of seawater?
Average ocean water has a salinity of 35g of dissolved salts. Expressed in %, it would be 96.5% pure water molecules and 3.5% of salts, dissolved gases, organic materials and undissolved particles.
What is the difference between tonicity and osmotic pressure?
Osmotic pressure is the pressure of a solution against a semipermeable membrane to prevent water from flowing inward across the membrane. Tonicity is the measure of this pressure. … Usually, there is a higher concentration of solutes on one side of the membrane than the other.