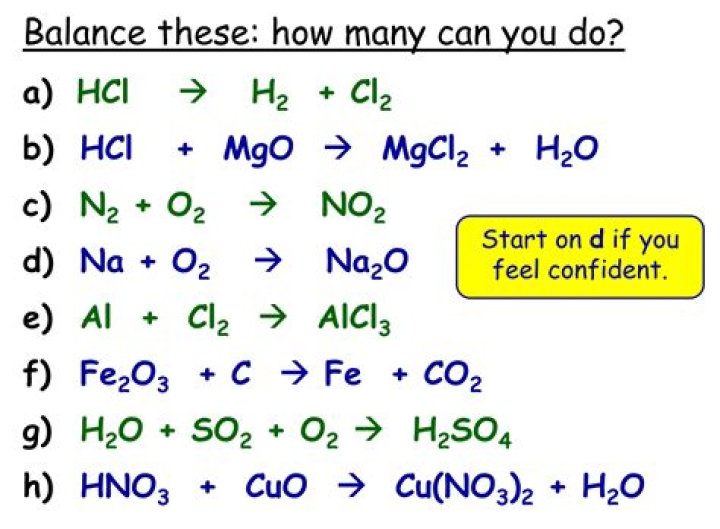
What law is the ultimate basis for a balanced chemical equation

Olivia Owen
Published Mar 14, 2026
The basis of balanced chemical equation is the law of conservation of mass. Mass can neither be created nor destroyed in a chemical reaction in ordinary chemical and physical changes.
What laws is the basis for balancing chemical equations?
Balancing equations demonstrates the fundamental law of the conservation of mass. It shows that you cannot create or destroy mass in a chemical reaction, so the mass stays constant.
Which law do we use to balance chemical reactions explain with an example?
Where there is a chemical reaction, there is a chemical equation. The chemical equation needs to be balanced so that it follows the law of conservation of mass. A balanced chemical equation occurs when the number of the different atoms of elements in the reactants side is equal to that of the products side.
What law is the ultimate basis for a correct chemical equation?
Chemical equation are balanced on the basis of The Law of Conservation of Mass. Mass of reactants equals mass of products. Therefore the number of Atoms of every element involved in the reaction remains same.What is chemical combination law?
The five laws of chemical combination are: Law of conservation of mass. Law of definite proportions. Law of multiple proportions. Gay Lussac’s law of gaseous volumes.
What is fundamental law of chemistry?
All matter consists of indivisible particles called atoms. Atoms of the same element are similar in shape and mass, but differ from the atoms of other elements. Atoms cannot be created or destroyed. Atoms of different elements may combine with each other in a fixed, simple, whole number ratios to form compound atoms.
What are the 3 laws of chemistry?
Laws of Chemistry The law of constant proportions. The law of multiple proportions. The law of reciprocal proportions.
How many laws are there in law of chemical combination?
There are five basic laws of chemical combinations that govern the chemical combinations of elements.What did Joseph Proust state regarding law of definite proportions?
The Law of Constant Composition, discovered by Joseph Proust, is also known as the Law of Definite Proportions. … The French chemist Joseph Proust stated this law the following way: “A chemical compound always contains the same elements combined together in the same proportion by mass.”
Which of the following laws of chemical combination is illustrated by the balanced chemical equation?Law of Conservation of Mass – As according to this law mass can neither be created nor destroyed. This is the reason why we always balance a chemical equation. Thus, for any chemical reaction or chemical change the mass of the reactants is equal to the mass of product formed.
Article first time published onWhat is an example of law in chemistry?
One of the most important laws in chemistry is the law of conservation of matter. This law states that matter can neither be created nor destroyed. … Biology, too, has laws that describe natural phenomena. One excellent example is Mendel’s law of segregation.
What is the scientific law?
In general, a scientific law is the description of an observed phenomenon. It doesn’t explain why the phenomenon exists or what causes it. The explanation of a phenomenon is called a scientific theory. It is a misconception that theories turn into laws with enough research.
What are the 4 laws of chemical combination?
- Law of Conservation of Mass. In simple terms, this law states that matter can neither be created nor destroyed. …
- Law of Definite Proportions. …
- Law of Multiple Proportions. …
- Gay Lussac’s Law of Gaseous Volumes. …
- Avogadro’s Law.
What are Daltons 3 laws?
This section explains the theories that Dalton used as a basis for his theory: (1) the Law of Conservation of Mass, (2) the Law of Constant Composition, (3) the Law of Multiple Proportions.
What is basic law matter?
According to the law of conservation of matter, matter is neither created nor destroyed, so we must have the same number and kind of atoms after the chemical change as were present before the chemical change.
What is the law of definite proportions and how does it apply to balancing chemical equation?
The law of definite proportions states that a given chemical compound always contains the same elements in the exact same proportion by mass.
What is chemical proportion law?
law of definite proportions, statement that every chemical compound contains fixed and constant proportions (by mass) of its constituent elements.
How does a balanced chemical equation agree with the law of definite proportions?
The Law of Definite Proportions states that a chemical compound will always have the same proportions or amount of each element by weight, no matter what the amount is, or source.
Which law is obeyed by this combination?
Answer: The law obeyed here is LAW OF MULTIPLE PROPORTIONS. Explanation: It states that if two elements can combine to form more than one compound, the masses of one element that combine with the fixed mass of the other element , are in the ratio of small whole numbers.
What is law of chemical combination class 11?
The law states that if two elements combine to form more than one compound, then the masses of one element that combines with the fixed mass of another element are in the ratio of small whole numbers.
What are the two laws of chemical combination class 9?
They are: Law of Conservation of Mass. Law of Constant Proportions.
What laws govern the formation of compounds?
Two laws—the law of definite proportions and the law of multiple proportions—describe the proportions in which elements combine to form compounds.
What are the basic laws of nature?
These fundamentals are called the Seven Natural Laws through which everyone and everything is governed. They are the laws of : Attraction, Polarity, Rhythm, Relativity, Cause and Effect, Gender/Gustation and Perpetual Transmutation of Energy. There is no priority or order or proper sequence to the numbers.
Are the laws of physics absolute?
In theory, the laws of physics are absolute. However, when it comes to the laws of thermodynamics —- the science that studies how heat and temperature relate to energy -— there are times where they no longer seem to apply.
How many scientific laws are there?
What are the five scientific laws? The five most popular scientific laws are Hooke’s Law of Elasticity, Archimedes’ Principle of Buoyancy, Dalton’s Law of Partial Pressures, Bernoulli’s Law of Fluid Dynamics and Fourier’s Law of Heat Conduction.
What law did John Dalton discover?
Dalton’s atomic theory was the first complete attempt to describe all matter in terms of atoms and their properties. Dalton based his theory on the law of conservation of mass and the law of constant composition. The first part of his theory states that all matter is made of atoms, which are indivisible.
What is Ernest Rutherford atomic theory?
Ernest Rutherford found that the atom is mostly empty space, with nearly all of its mass concentrated in a tiny central nucleus. The nucleus is positively charged and surrounded at a great distance by the negatively charged electrons.