What measurement describes the pressure of a gas

Rachel Hunter
Published Apr 13, 2026
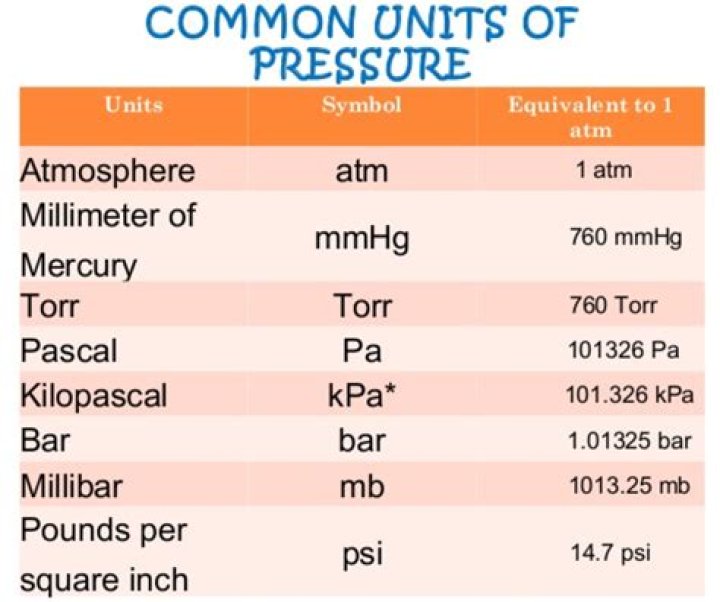
The pressure of a gas may be expressed in the SI unit of pascal or kilopascal, as well as in many other units including torr, atmosphere, and bar. Atmospheric pressure is measured using a barometer; other gas pressures can be measured using one of several types of manometers.
What describes the pressure of a gas?
PRESSURE is a force exerted by the substance per unit area on another substance. The pressure of a gas is the force that the gas exerts on the walls of its container.
What is the measurement of gas?
Gas is sometimes measured in cubic feet at a temperature of 60 degrees Fahrenheit and an atmospheric pressure of 14.7 pounds per square inch. Gas production from wells is discussed in terms of thousands or millions of cubic feet (Mcf and MMcf). Resources and reserves are calculated in trillions of cubic feet (Tcf).
What measurement describes pressure?
The SI unit for pressure is the pascal (Pa), equal to one newton per square metre (N·m−2 or kg·m−1·s−2). This special name for the unit was added in 1971; before that, pressure in SI was expressed in units such as N·m−2.What is the unit of pressure of gas?
The pascal (Pa) is the standard unit of pressure. A pascal is a very small amount of pressure, so the more useful unit for everyday gas pressures is the kilopascal (kPa). A kilopascal is equal to 1000 pascals. Another commonly used unit of pressure is the atmosphere (atm).
How is natural gas pressure measured?
How do you measure the gas pressure? You will need an instrument called a Manometer. This tool allows you to measure the pressure of gas in the system. Manometers are available that measures a specific range of pressure in inches of water column or pounds per square inch.
How gas pressure is produced in gas tank?
Gas pressure is caused when gas particles hit the walls of their container. The more often the particles hit the walls, and the faster they are moving when they do this, the higher the pressure.
What is the temperature of a gas a measure of?
The temperature of a gas is a measure of the average translational kinetic energy of the molecules.How do you measure gas in a lab?
The simplest method Is displacement of water inside an Inverted graduated measuring cylinder or graduated test tube placed inside a water bath. This method Is suitable for gases such as Hydrogen or Nitrogen but does not work for water soluble gases such as ammonia or chlorine.
How do u calculate pressure?- To calculate pressure, you need to know two things:
- Pressure is calculated using this equation:
- pressure = force ÷ area.
What uses gas pressure?
An example of an application that uses gas pressure for transportation is the engine of a car. Gasoline or diesel fuel is added to air and compressed in the engine.
How much pressure is in a natural gas line?
The natural gas pressure of the gas line leading to the home ranges from approximately 1/4 psi to 60 psi, depending on the number of homes or businesses served by the line. This compares to pressures of up to 1,500 psi for large-volume pipelines used to move the gas from the well fields to the local utilities.
How do you check gas pressure?
- Step 1 – Turn on appliance. Turn on one appliance (usually the boiler). The gas should be on full.
- Step 2 – Read Gas Test Gauge. Take a reading from the gas test gauge. The working pressure of most meters should be between 19 mbar and 23 mbar.
How is natural gas flow measured?
A flow meter is used to measure gas flow and should demonstrate high accuracy, low-pressure drop, and direct mass flow readings. High-quality natural gas measurement increases efficiency in boiler/burner control, fuel-to-air ratio, and custody transfer applications.
How much pressure is in a gas cylinder?
The regular LPG cylinders have pressure from 5.5kg/sqcm to 6.5kg/sqcm depending on the surrounding temperature. A new sealed LPG cylinder has approx. 85% liquid and rest vapor.
How much gas is in a cylinder?
Cooking gas cylinder weight is actually a net 14.2 kg LPG gas cylinder, if we are talking about India. To calculate volume, in litres, takes some simple arithmetic. With a 75:25 butane:propane mix, 1kg = 1.783L. Therefore, there is 25.34L in a 14.2 kg LPG gas cylinder.
How do you measure gas composition?
Gas composition can be determined by in situ direct measurement using portable gas sensors. These need frequent calibration based on several gas standards covering the range of interest. Relative uncertainties in readings are generally less than 5%.
What instrument measures volume of gas?
A eudiometer is a laboratory device that measures the change in volume of a gas mixture following a physical or chemical change.
How do you describe gas?
Gas is a state of matter that has no fixed shape and no fixed volume. … The particles move very fast and collide into one another, causing them to diffuse, or spread out, until they are evenly distributed throughout the volume of the container.
How does the temperature of a certain amount of gas affect its pressure?
The temperature of the gas is proportional to the average kinetic energy of its molecules. Faster moving particles will collide with the container walls more frequently and with greater force. This causes the force on the walls of the container to increase and so the pressure increases.
What is the volume of the gas at standard temperature and pressure?
At Standard Temperature and Pressure (STP), 1 mole of any gas will occupy a volume of 22.4 L.
How do you find the pressure of a gas in physics?
Calculate the pressure when the volume increases to 8.00 × 10 -6 m 3 at a constant temperature. Pressure and volume of a given mass of gas at a constant temperature can be given by: P 1 × V 1 = P 2 × V 2 (where P 1 and V 1 are the pressure and temperature at the start, and P 2 and V 2 are at the end).