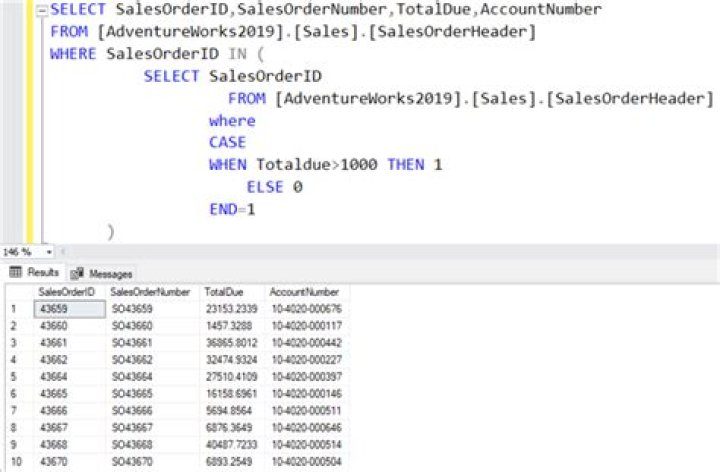
What procedures must be described in an agreement called

Lily Fisher
Published May 01, 2026
These procedures must be described in an agreement called an “assurance of compliance” with a U.S. federal agency.
What procedures must be described in an agreement called an Assurance of?
What procedures must be described in an agreement called an “assurance of compliance” with OHRP? procedures in place that ensure that subjects will be protected in a manner commensurate with the Common Rule, including review by an independent committee comparable to an IRB.
What is the term for management controls that are built in to a research study?
What is the term for management controls that are built in to a research study (for example, independent data analysis)? Inherent controls.
What activities constitutes engagement in research?
Which of the following activities constitutes engagement in research? Obtaining informed consent and conducting research interviews. Laws, customs, and norms in the area in which the research will be conducted.Which of the following statements most accurately describes the requirement for the documentation of minors assent?
Which of the following statements most accurately describes the requirement for the documentation of minors’ assent to participate in research? Documentation is required unless waived by an IRB.
Which of the following are the three principles discussed in the Belmont Report?
The three principles discussed in the Belmont Report are Respect for Persons, Beneficence, Justice.
What statement is correct about informed consent?
Which statement is correct about informed consent? The informed consent document, regardless of the research topic, must always include the following: A statement describing the extent, if any, to which confidentiality of records identifying the subject will be maintained.
What is engaged in research?
Engage is a premier market research firm specializing in nationwide recruiting and field management. … Our field management division is comprised of seasoned professionals who have formed strategic relationships with national facilities.What does engagement mean in research?
In brief, an “engaged” institution is one whose agents (faculty, students, or staff) recruit and secure consent from subjects, conduct research procedures, or who receive or share private, identifiable information. … This definition parallels the federal definition of research with human subjects.
Who has the authority to determine whether research is exempt from requirements described in the HHS regulations including IRB oversight?OHRP notes that the HHS retains final authority as to whether a particular human subjects research study conducted or supported by HHS is exempt from the HHS regulations (45 CFR 46.101(c)).
Article first time published onWhat is an example of an institutional COI?
Examples of institutional financial interests include: … Covered Official relationships where an institutional official receives payments, honoraria, royalties (including those from U-M), equity, options and warrants, company positions (e.g., board directorships and/or management), or gifts.
What is a COI management plan?
Conflict of Interest (COI) Management Plan Outline Describe the external relationship [Significant Financial Interest Disclosure form] Identify areas of potential COI [data integrity; undue influence on students/postdocs] Develop strategies to guard against COI affecting data or behavior and draft in a plan.
What is individual financial COI?
An individual COI may arise when an individual has a personal or financial interest, which may affect or appear to affect the design, conduct, or reporting of the research.
Which of the following statements is true regarding the responsibilities of reviewers quizlet?
Which of the following statements is true regarding the responsibilities of a reviewer? A reviewer’s conflict of interest should be disclosed to the journal editor or grant agency. Reviewers have a responsibility to promote ethical peer review by: Preserving the confidentiality of the submission.
Which federal regulation or law governs?
Which federal regulation or law governs how researchers can obtain data about subjects’ disciplinary status in school from academic records? The Family Educational Rights and Privacy Act.
Which of the following studies would need IRB approval?
Which of the following studies need IRB approval? Studies collecting data about living individuals.
What are the 4 elements of informed consent?
There are 4 components of informed consent including decision capacity, documentation of consent, disclosure, and competency.
Which of the following statements describes the informed consent requirements in the US?
Which of the following statements describes the informed consent requirements in the U.S.? Subjects must be given sufficient time to decide whether or not to participate. An investigator calls you to get your opinion on whether an investigational device may be used in an individual with a life-threatening condition.
What is considered informed consent and how is obtained?
The process of informed consent occurs when communication between a patient and physician results in the patient’s authorization or agreement to undergo a specific medical intervention.
Which of the following best describes the principle of informed consent as described in the Belmont?
Determining that the study has maximized benefits and minimized risks. Which of the following best describes the principle of informed consent as described in the Belmont Report? Information, comprehension, voluntariness.
Which of the following best describes the principle of respect for persons as described in the Belmont Report?
The Belmont Report states that “respect for persons incorporates at least two ethical convictions: first, that individuals should be treated as autonomous agents, and second, that persons with diminished autonomy are entitled to protection.
Which of the following best describes the principle of respect for persons as described in the Belmont Report ?\?
The principle of respect for persons is interpreted to mean that researchers should, if possible, receive informed consent from participants, and the Belmont Report identifies three elements of informed consent: information, comprehension, and voluntariness.
How is public engagement defined?
Our definition “Public engagement describes the myriad of ways in which the activity and benefits of higher education and research can be shared with the public. Engagement is by definition a two-way process, involving interaction and listening, with the goal of generating mutual benefit.”
How is community engagement be conducted in the community?
Community Engagement is…the process of working collaboratively with and through groups of people affiliated by geographic proximity, special interest, or similar situations to address issues affecting the well-being of those people It is a powerful vehicle for bringing about environmental and behavioral changes that …
What is a public engagement strategy?
A Public Engagement Plan (PEP) outlines how to involve the public in a planning project. It describes goals and objectives and identifies specific approaches and tools. The PEP can be an internal document, or can be made public to educate citizens about local government plans for outreach and dialogue.
What is institutional engagement?
Institutional Engagement University resources intentionally offered without undue barriers to the community. … Examples include making Briggs library cards available for community members, making campus events accessible, and choosing to use local and sustainable businesses to supply services or goods.
Why do we engage in research?
Research allows you to pursue your interests, to learn something new, to hone your problem-solving skills and to challenge yourself in new ways.
Why is community engaged research important?
A community-engaged research study may incorporate both qualitative and quantitative methods. This approach encourages recognition of the strengths of community institutions and individuals and encourages people and groups to build on those strengths.
What is determination for exemption?
Studies that receive an exemption determination from IRB are exempt from the specific regulations and requirements in Title 45, Part 46 of the Code of Federal Regulations. Please note, however, that they are still considered human subject research.
Who determines exempt research?
A determination of exemption must be made by the JHM IRBs and principal investigators must submit all such studies for their review. Although the research may qualify as exempt, it must still be conducted in accordance with the ethical principles for human subjects research outlined in the Belmont Report. 1.
Which Belmont principle requires fair procedures in the selection of research participants?
— Just as the principle of respect for persons finds expression in the requirements for consent, and the principle of beneficence in risk/benefit assessment, the principle of justice gives rise to moral requirements that there be fair procedures and outcomes in the selection of research subjects.