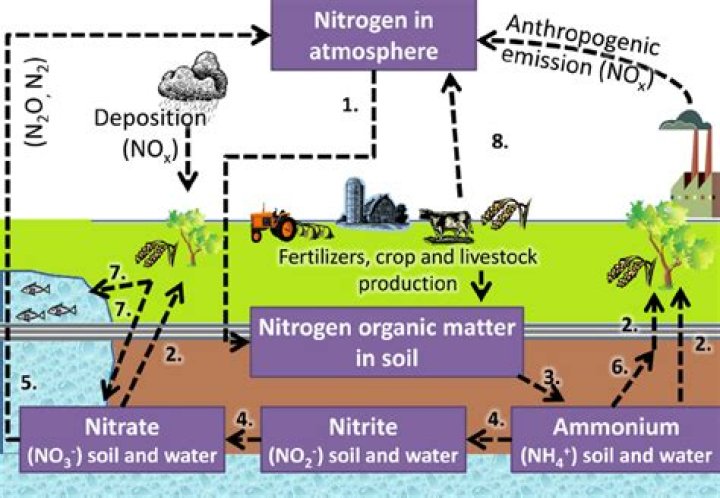
Where does nitrate in soil come from

Olivia Owen
Published Mar 23, 2026
Nitrate (NO3) is a naturally occurring form of nitrogen in soil. This form of nitrogen is created when nitrification, the conversion of ammonium into nitrate, occurs. Nitrate is used as food by plants for growth and production.
Where does soil nitrogen come from?
Nitrogen is added to soil naturally from N fixation by soil bacteria and legumes and through atmospheric deposition in rainfall. Additional N is typically supplied to the crop by fertilizers, manure, or other organic materials.
Where do nitrates come from in plants?
Nitrate (NO3−) is the most labile form of N. Nitrates may originate from manure, the nitrification of plants and other organic matters, or from synthetic fertilisers.
Where do nitrates come from?
Nitrate is a chemical found in most fertilizers, manure, and liquid waste discharged from septic tanks. Natural bacteria in soil can convert nitrogen into nitrate. Rain or irrigation water can carry nitrate down through the soil into groundwater.How is nitrogen made?
Nitrogen is produced commercially almost exclusively from air, most commonly by the fractional distillation of liquid air. … In another approach, passing ammonia gas over a hot metallic oxide will result in the formation of free nitrogen, the free metal, and water.
How nitrates are formed?
Nitrates are produced by a number of species of nitrifying bacteria in the natural environment using ammonia or urea as a source of nitrogen. Nitrate compounds for gunpowder were historically produced, in the absence of mineral nitrate sources, by means of various fermentation processes using urine and dung.
Where is nitrate found in the environment?
Because nitrite is easily oxidized to nitrate, nitrate is the compound predominantly found in groundwater and surface waters. Contamination with nitrogen containing fertilizers (e.g. potassium nitrate and ammonium nitrate), or animal or human organic wastes, can raise the concentration of nitrate in water.
Why is nitrate good for plants?
All nitrates add nitrogen to the soil. … Both nitrate ions and ammonium ions can be taken up by plants (some prefer one to the other). These ions provide nitrogen for the plant to make its own amino acids for protein synthesis. Except in very high nitrogen soils, the addition of extra nitrate will stimulate plant growth.Do plants need nitrate?
Plant roots absorb mineral salts including nitrates needed for healthy growth. For healthy growth plants need mineral ions including: – Nitrate for producing amino acids which are then used to form proteins. – Magnesium which is needed for chlorophyll production.
Where is nitrogen found in nature?Nitrogen is in the soil under our feet, in the water we drink, and in the air we breathe. In fact, nitrogen is the most abundant element in Earth’s atmosphere: approximately 78% of the atmosphere is nitrogen! Nitrogen is important to all living things, including us.
Article first time published onHow is nitrogen produced for fertilizer?
Ammonia in water, known as aqua ammonia, is free to escape into the air and, therefore, when used as a nitrogen fertilizer, must be injected under the soil surface. … Then during the growing season, soil microorganisms convert the ammonium to nitrate, which is the main form taken up by plants.
What is the source of nitrogen in fertilizer?
Fifty percent of the total nitrogen comes from urea, the other 50 percent from ammonium nitrate, resulting in 25 percent NO3-, 25 percent NH4+ and 50 percent urea. Can be blended with other nutrients and many agricultural chemicals.
What do nitrates cause?
Nitrate can affect how our blood carries oxygen. Nitrate can turn hemoglobin (the protein in blood that carries oxygen) into methemoglobin . High levels can turn skin to a bluish or gray color and cause more serious health effects like weakness, excess heart rate, fatigue, and dizziness.
How is nitrate made in nature?
In nature, nitrate and nitrite can be found in igneous and volcanic rocks. Nitrate and nitrite salts completely dissolve in water. Bacteria in soil and plants use oxygen to change nitrite into more stable nitrate, which can be converted back to nitrite by other bacteria when oxygen is lacking.
Are nitrates acidic?
An electrostatic potential map of the nitrate ion. Areas colored red are lower in energy than areas colored yellow. In inorganic chemistry, a nitrate is a salt of nitric acid characterized by a negatively charged ion composed of one nitrogen atom bound to three oxygen atoms.
What are examples of nitrates?
- nitroglycerin sublingual tablet (Nitrostat)
- nitroglycerin lingual aerosol (Nitromist)
- nitrolinglycerin pumpspray (Nitrolingual Pumpsprapy)
- nitroglycerin lingual (Nitrolingual Pumpspray)
- nitroglycerin transdermal ointment (Nitro-Bid)
What happens if a plant lacks nitrate?
Nitrate deficiency Nitrates are present in high levels in plant fertilisers. Without nitrates, the amount of chlorophyll in leaves reduces. This means leaves turn a pale green or yellow colour. This reduces the plant’s ability to photosynthesise and grow properly, which reduces the farmers’ crop yield .
How does nitrates affect plant growth?
Nitrogen, potassium and phosphorus are the three major nutrients plants need to grow successfully. In particular, nitrogen is responsible for foliage development. Plants use nitrogen in the form of nitrates, which is nitrogen mixed with oxygen. They usually absorb nitrates from the soil.
Why are nitrates used in fertilizers?
Nitrates are chiefly produced to be used as fertilizers in agriculture because they have high solubility and biodegradability. The main nitrate fertilizers are ammonium, sodium, potassium, and calcium salts.
What happens when there is too much nitrate in soil?
Increase in nitrate, like increase in any salt will increase the osmotic concentration of the soil solution. … If the solution outside gets too concentrated, there will come a point where the plant is not able to take up any water against the concentration gradient and the plant will start to wilt.
Is nitrate bad for soil?
Soil Tests and Fertilization When fertilizing, take care to avoid excessive nitrate levels because they can lead to groundwater contamination and/or nitrate poisoning of grazing animals.
Are nitrates toxic to plants?
IT is generally believed that nitrite is an intermediate product in the conversion of ammonium to nitrate in the soil where the conversion of nitrite to nitrate is important since relatively small quantities may have toxic effects on plant growth.
What plants add nitrogen to the soil?
Legumes such as peas, peanuts, beans, clover, and alfalfa are the best plants for adding nitrogen to soil. According to Wikipedia, a legume is a plant that has “symbiotic nitrogen-fixing bacteria in structures called root nodules.” (The specific type of bacteria is called Rhizobia).
What are 3 sinks of nitrogen?
Nitrogen import into sinks. During the reproductive phase, seeds are major N sinks in annual plants, while during vegetative growth and in perennials, roots, developing leaves, and stems or trunks are strong sinks for N.
How was nitrogen discovered?
Nitrogen was discovered in 1772 by chemist and physician Daniel Rutherford, when he removed oxygen and carbon dioxide from air, demonstrating that the residual gas would not support living organisms or combustion, according to the Los Alamos National Laboratory.
How do you make phosphorus rich fertilizer?
Dig up 2 teaspoons of soil from the garden and add ½ cup of vinegar. If the soil beings to fizz, then it’s on the alkaline side, with a pH of between 7 and 8. The calcium and phosphorus bond is stable, so it won’t burn the plants as other fertilizers will if too much is added to the soil.
Which fertilizer produces acidity in soil?
Ammonium-based fertilisers are the major contributors to soil acidification, especially if the nitrogen is leached rather than taken up by plants.
How can I add nitrogen to my soil naturally?
- Add Composted Manure.
- Use a Green Manure Crop.
- Plant Nitrogen-Fixing Plants.
- Mix Coffee Grounds in the Soil.
- Use Fish Emulsion.
- Spread Grass Clippings As Mulch.
- Use an Actual Plant Fertilizer.
What is the best source of nitrogen?
- Green Manure: Cover crops – such as alfalfa, clover, peas, and other legumes – are able to absorb nitrogen from the air and release it into the soil. …
- Soybean Meal: Similar to cottonseed meal, soybean meal is a slow release source of nitrogen made from ground soybeans.
Why are nitrates bad?
Sodium nitrate, a preservative that’s used in some processed meats, such as bacon, jerky and luncheon meats, could increase your heart disease risk. It’s thought that sodium nitrate may damage your blood vessels, making your arteries more likely to harden and narrow, leading to heart disease.
Why are nitrates bad for the environment?
The environmental impacts of nitrate pollution include ecological and amenity damage to our rivers, lakes and coasts; higher costs, energy use and carbon emissions for drinking water treatment and long-term impacts for our groundwater.



