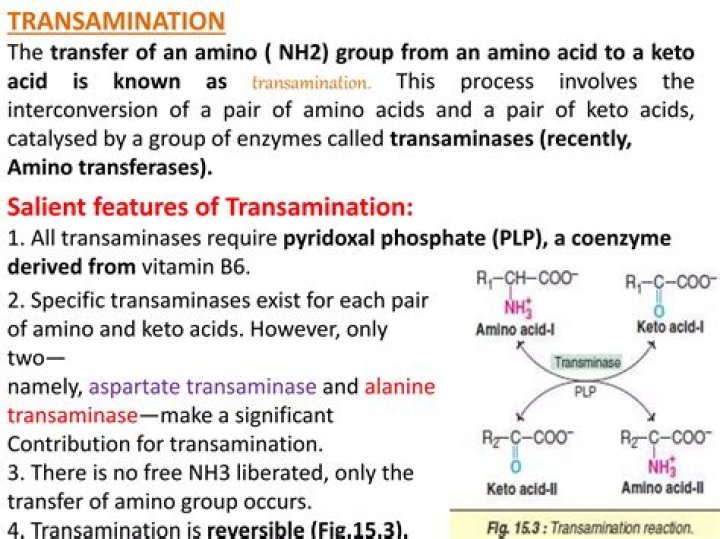
Where does transamination of amino acids occur

Sarah Rodriguez
Published Apr 03, 2026
The liver is the principal site of amino acid metabolism, but other tissues, such as the kidney, the small intestine, muscles, and adipose tissue, take part. Generally, the first step in the breakdown of amino acids is the separation of the amino group from the carbon skeleton, usually by a transamination reaction.
Where does Transamination take place in the cell?
Transamination and deamination take place in the cytoplasm of all the cells.
Where does deamination and transamination primarily occur in the body?
In the human body, deamination takes place primarily in the liver, however it can also occur in the kidney. In situations of excess protein intake, deamination is used to break down amino acids for energy. The amino group is removed from the amino acid and converted to ammonia.
What is the site of Transamination?
Transamination reactions combine reversible amination and deamination, and they mediate redistribution of amino groups among amino acids. Transaminases (aminotransferases) are widely distributed in human tissues and are particularly active in heart muscle, liver, skeletal muscle, and kidney.Is transamination anabolic or catabolic?
All amino acids except, lysine, threonine, proline & hydroxyproline participate in transamination. It involves both anabolism & catabolism, since – reversible.
Do all amino acids undergo transamination?
All of the amino acids except lysine, threonine, proline, and hydroxyproline participate in transamination reactions. Transaminases exist for histidine, serine, phenylalanine, and methionine, but the major pathways of their metabolism do not involve transamination.
What is the role of transamination in amino acid biosynthesis?
Transamination is the process by which amino groups are removed from amino acids and transferred to acceptor keto-acids to generate the amino acid version of the keto-acid and the keto-acid version of the original amino acid.
Which amino acids do not undergo transamination?
Being a major degradative aminoacid pathway, lysine, proline and threonine are the only three amino acids that do not always undergo transamination and rather use respective dehydrogenase.When amino acids bind a transaminase enzyme their amino group is transferred?
When amino acids bind a transaminase enzyme, their amino group is transferred to associated with the transaminase before it is transferred to an alpha-keto acid.
What is transamination and example?Transamination as the name implies, refers to the transfer of an amine group from one molecule to another. This reaction is catalyzed by a family of enzymes called transaminases. … A specific example is the transamination of alanine to make pyruvic acid and glutamic acid.
Article first time published onWhich cofactor is essential to all transamination reactions?
Pyridoxal phosphate acts as a coenzyme in all transamination reactions, and in some oxylation and deamination reactions of amino acids.
Which of the following is an example of a transamination process?
An example of a transamination process is glutamate = hexanoic acid +
What is transamination give its significance?
Transamination is a biologically important process by which living cells exchange the oxygen atoms of α-oxoacids with amino groups (and associated hydrogen atoms) of amino acids such as glutamic acid (Figures 1 and 2).
What species is formed from the transamination and oxidative deamination of the amino group in an amino acid?
The amino acid is transaminated to produce a molecule of glutamate. Glutamate is the one amino acid that undergoes oxidative deamination to liberate free ammonia for the synthesis of urea. Once free ammonia is formed in peripheral tissues, it must be transferred to the liver for the conversation to urea.
What is the significance of transamination and deamination reactions?
Transamination reactions are responsible for the synthesis of nonessential amino acids. In comparison, deamination is a biochemical reaction responsible for the breakdown of excess proteins in the liver.
How does transamination reaction Help nitrogen transfer?
Because transamination reactions are reversible they can be used to remove nitrogen from amino acids or to transfer nitrogen to α-keto acids to form amino acids. They participate in both amino acid degradation and amino acid synthesis.
What is transamination in plants?
Transamination represents a class of reaction wherein the amino nitrogen of an amino acid (donor) is transferred to aminate the carbonyl group of a keto acid (acceptor). … The role of transamination in amino acid synthesis is discussed in the chapter “The synthesis of amino acids in plants”, p. 224.
Does Transamination occur in cytosol?
Valine, leucine, and isoleucine are branched-chain amino acids (BCAAs) and their degradation pathways are predominantly localized in mitochondria except the first transamination step, which occurs in cytoplasm (8).
Which of these amino acids are converted to alpha ketoglutarate?
Alpha-ketoglutarate is produced from arginine, glutamate, glutamine, histidine and proline.
Which of the following cofactor is essential for the activity of acetyl CoA carboxylase?
d-(+)-Biotin is a cofactor designed for the transfer of CO2 in carboxylase and transcarboxylase enzymes, such as acetyl-CoA carboxylase-α, acetyl-CoA carboxylase-β, and methylcrotonyl-CoA, propionyl-CoA, and pyruvate carboxylases.
Is transamination the same as deamination?
The key difference between transamination and deamination is that the transamination is the transfer of an amino group to a keto whereas the deamination is the removal of an amino group. Transamination and deamination are two types of chemical reactions in which the change of amino groups in organic molecules occur.



