Where is ethane found in nature

Rachel Hunter
Published Mar 03, 2026
Ethane (C2H6) is present in nature in petroleum gas and as a minor component in natural gas (between 1% and 7%). Pyrolysis of ethane is used extensively for the production of ethylene (C2H4).
Where can you find ethane?
Ethane is structurally the simplest hydrocarbon that contains a single carbon–carbon bond. The second most important constituent of natural gas, it also occurs dissolved in petroleum oils and as a by-product of oil refinery operations and of the carbonization of coal.
What is ethane used for in everyday life?
Ethane is mainly used to produce ethylene, a feedstock to make plastics. Ethane is mainly used to produce ethylene, which is then used by the petrochemical industry to produce a range of intermediate products, most of which are converted into plastics.
Is ethane natural?
Ethane is the most prevalent natural gas liquid, or NGL, and used solely as a feedstock for petrochemicals. In oil-rich areas such as the Permian, the NGLs flow out of wells as liquids in the stream of crude.Is ethane natural or synthetic?
EthaneFlash point-135 °CRelated CompoundsRelated alkanesMethane; propane
How does ethane affect the environment?
A component of natural gas, ethane plays an important role in Earth’s atmosphere. As it breaks down near Earth’s surface it can create ground-based ozone pollution, a health and environmental risk. … There is high interest by scientists in methane since it is a strong greenhouse gas, said Helmig.
Is ethane part of natural gas?
Natural gas is a hydrocarbon mixture consisting primarily of saturated light paraffins such as methane and ethane, both of which are gaseous under atmospheric conditions. The mixture also may contain other hydrocarbons, such as propane, butane, pentane, and hexane.
What type of hydrocarbon is ethene?
Ethylene, or ethene, is an unsaturated hydrocarbon. It is a colorless gas. Its chemical formula is C2H4 where there is a double bond between the carbons. It is extremely flammable and it has planar structure.Is ethane organic or inorganic?
Hydrocarbons are very common and familiar organic compounds. Gasoline is a hydrocarbon; so too are methane, ethane, propane and butane.
How is ethane produce?How Ethane Is Produced. When first produced, oil and natural gas must be processed to remove various impurities. Natural gas processing removes hydrocarbons like ethane, butane, propane and other hydrocarbons, as well as water and other impurities from the gas stream.
Article first time published onWhat is the common name for ethane?
1. Ethane; common name is bimethyl or dimethyl.
Is ethane a greenhouse gas?
Ethane, although it will act as a greenhouse gas, only stays in the atmosphere for a few months before breaking down into other compounds, rather than the 10 years that methane remains in the atmosphere. … The researchers used ethane-to-methane ratios from oil and gas production basins for this study.
How many atoms are there in ethane?
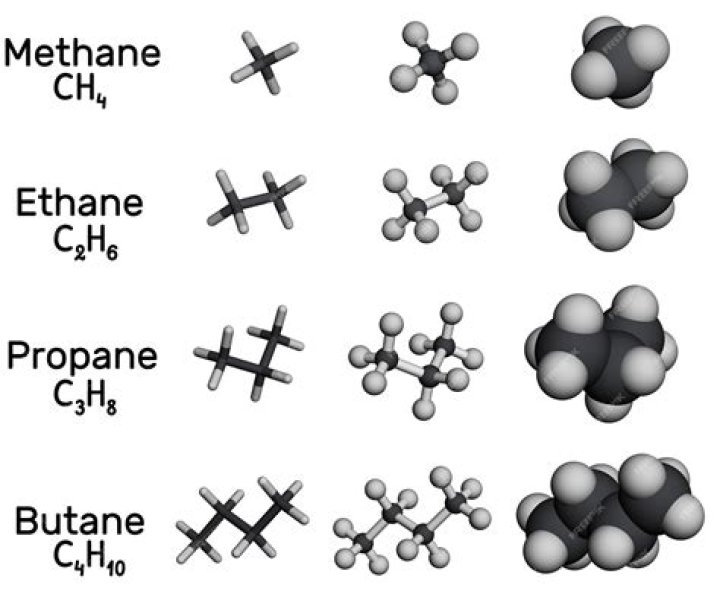
Ethane is a colorless, odorless, and flammable gas with a chemical formula of C2H6; it has two carbon (C) atoms and six hydrogen (H) atoms.
Is ethane ionic or covalent?
Ethane, C2H6, is a hydrocarbon molecule, an odorless gas at room temperature. The covalent bonding picture of ethane is very similar to that of methane above. Two carbon atoms, each carrying four valence electrons, come together to share a pair of electrons. We’ll start calling that a single bond (or sigma bond).
Is NGL natural gas?
Natural gas liquids (NGLs) are hydrocarbons—in the same family of molecules as natural gas and crude oil, composed exclusively of carbon and hydrogen. Ethane, propane, butane, isobutane, and pentane are all NGLs (see table above). There are many uses for NGLs, spanning nearly all sectors of the economy.
Is ethane a natural gas liquid?
Natural gas liquids (NGLs) are a group of hydrocarbons that includes ethane, propane, butane, isobutane, and natural gasoline. NGLs are differentiated from one another by the number of carbon atoms in their molecular chain.
What is wet natural gas?
wet gas, natural gas that contains an appreciable proportion of hydrocarbon compounds heavier than methane (e.g., ethane, propane, and butane). … Alternatively, the propane and other lighter compounds may be marketed as liquefied petroleum gas (LPG), and heavier hydrocarbons may be made into gasoline (petrol).
How is ethene extracted from the earth?
Ethene is produced from the cracking of fractions obtained from distillation of natural gas and oil. (which can vary considerably), and what other products from cracking are needed. The vast majority of ethene is produced by steam cracking. … The US produces about 25 million tonnes of ethene a year.
Is ethylene environmentally friendly?
Ethylene sourced from sugarcane is claimed to be greener than that produced from fossil fuel-based sources [7]. … However, in the longer term, bio-based feedstocks are the only sustainable option for producing chemicals. The cracking of naphtha or of ethane to ethylene is highly energy intensive [9].
What is ch3 in chemistry?
A methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms — CH3. In formulas, the group is often abbreviated Me. Such hydrocarbon groups occur in many organic compounds. It is a very stable group in most molecules.
What does C2H6 mean in chemistry?
C2H6, 2 atoms of carbon combine with 6 atoms of hydrogen to form ethane.
How is ethene different from ethane?
Ethane is an alkane and as such is saturated, meaning it has all single bonds. Ethene is an alkene and as such is unsaturated, meaning it has one carbon to carbon double bond. … There is no visible sign of reaction when bromine is added to an alkane.
What is the state of ethene?
NamesAppearancecolorless gasDensity1.178 kg/m3 at 15 °C, gasMelting point−169.2 °C (−272.6 °F; 104.0 K)Boiling point−103.7 °C (−154.7 °F; 169.5 K)
Where is ethylene produced?
Ethylene is produced from essentially all parts of higher plants, including leaves, stems, roots, flowers, fruits, tubers, and seeds.
What substance found in the earth do we obtain ethene from?
However, ethene is made from crude oil, which is a non-renewable resource.
How is ethane stored?
Large volumes of NGLs are primarily stored as a pressurized liquid in underground caverns, but some areas without suitable geology may use aboveground tanks.
Why is ethene called ethylene?
Ethene is the formal IUPAC name for H2C=CH2, but it also goes by a common name: Ethylene. The name Ethylene is used because it is like an ethyl group (CH2CH3) but there is a double bond between the two carbon atoms in it.
How is ethane produced from natural gas?
The main technique to extract ethane from natural gas is low-temperature separation, that is, cooling of the gas stream until a liquid fraction containing the bulk of the ethane and other gas liquid components is condensed. … The oil used is in the gas-oil distillation range and is usually refrigerated.
What type of solid is ethane?
Ethane (chemical formula C2H6) is a colorless, odorless, combustible gas at standard conditions (temperature 0°C and pressure 1.01325 bar). Because of its molecular structure – which consists of two combined methyl groups – ethane is also sometimes called bimethyl or dimethyl. …
Is ethane an acid or base?
Thus, the methoxide anion is the most stable (lowest energy, least basic) of the three conjugate bases, and the ethyl carbanion anion is the least stable (highest energy, most basic). Conversely, ethanol is the strongest acid, and ethane the weakest acid.
Is ethane lighter than air?
Ethane appears as a colorless odorless gas. It is easily ignited. The vapors are heavier than air.