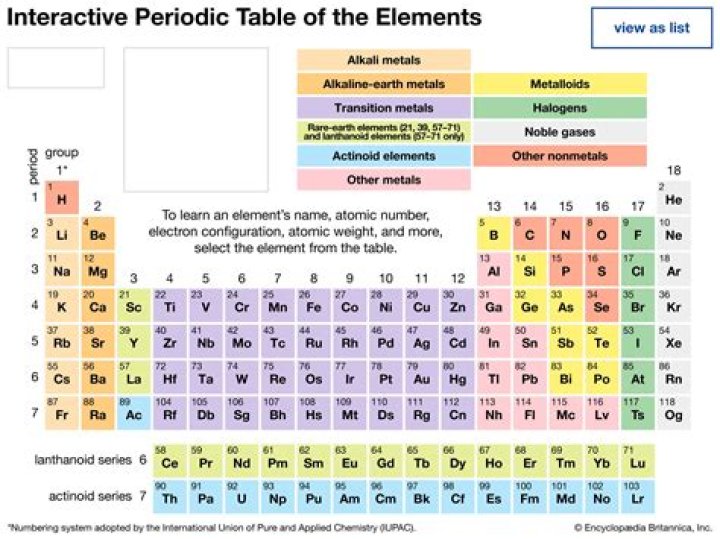
Which group 2 element is most metallic in character

Sarah Rodriguez
Published May 08, 2026
Among the group 2 elements, radium is known to have the greatest metallic character and it can, therefore, be considered the most metallic group 2 element. This is because the metallic character of elements increases while traversing down a group, and radium is at the bottom of group 2 in the modern periodic table.
Which element is most metallic in character?
The most metallic element is francium. However, francium is a man-made element, except for one isotope, and all isotopes are so radioactive they almost instantly decay into another element. The natural element with the highest metallic character is cesium, which is found directly above francium on the periodic table.
Which element is most metallic in group 15?
Out of the group, bismuth has the lowest electronegativity and ionization energy, which means that it is more likely to lose an electron than the rest of the Group 15 elements. This is why bismuth is the most metallic of Group 15.
What metal group has the greatest metallic character?
Group 1 is the one on the periodic table which has the greatest metallic character and group 17 is the one with the lowest metallic character.Which element has the most metallic character in group 13?
Boron belongs to the 13 group of a periodic table. Aluminium, indium and thallium are the members of the boron family. Thallium is the biggest in size among all so we can conclude that it has the highest metallic character.
Which of the following is metallic element?
Examples of metals are Lithium, Beryllium, Sodium, Chromium, Nickel, Gold, Copper, Silver, Iron and Zinc. Note: Metals generally have many uses including machinery, automobiles, aeronautics, trains, satellites, Industries, cooking utensils, water boilers etc.
Which element has the least metallic characteristics?
The least metallic or most non-metallic element is fluorine. Halogens near the top of the periodic table are the least metallic elements, not the noble gases. So, fluorine is more electronegative than helium or neon, even though these noble gases are to the right of fluorine on the table.
Which element is in Group 2 2A and Period 3?
31ANa2AMg4ASiWhat is metallic character in periodic table?
Metallic character refers to the level of reactivity of a metal. Non-metallic character relates to the tendency to accept electrons during chemical reactions. Metallic tendency increases going down a group. Non-metallic tendency increases going from left to right across the periodic table.
Which is the most metallic element in period 3?Textbook solution. The most metallic element is sodium i.e., Na and the most non-metallic element is chlorine i.e., Cl.
Article first time published onWhich is more metallic aluminum or gallium?
However, gallium is an exception. The atomic radius i.e., the metallic radius of gallium (135 pm) is less than that of aluminium (143 pm).
Which is more metallic B or C Why?
The first ionisation enthalpy of C is more than B on account of higher nuclear charge. … The order of first ionisation enthalpy among these elements is : Al < Si < B < C (ii) Aluminium is the most metallic element among the elements B, Al, C and Si.
Which is the highest non metallic character?
Fluorine has the highest non – metallic character as on going down through the 7th group in a periodic table, electronegativity gradually decreases.
Which element is most non metallic?
The most non-metallic element in the periodic table is Fluorine.
Which group has least metallic character?
Group 17 and group 18 have the least or lowest metallic character.
Which group of elements is least metallic?
If we look at the periodic table group 17 and group 18 have the least or lowest metallic character.
Which is least metallic in the nitrogen group?
The two least metallic elements of the heavier pnicogens, phosphorus and arsenic, form very stable oxides with the formula E4O10 in the +5 oxidation state (part (b) in Figure 22.5. 2. In contrast, Bi2O5 is so unstable that there is no absolute proof it exists.
Why does metallic character increase down a group?
As we move down the group, the number of shells increases. The effective nuclear charge experienced by valence electrons decreases because the outermost electrons move farther away from the nucleus. … By losing the electrons, element gains positive charge. Hence, metallic character increases down the group.
What are the two most common metals found in the earth's crust?
RankElementAbundance in crust (ppm) by source1Oxygen461,0002Silicon282,0003Aluminium82,3004Iron56,300
Which element of Group 2 is non metallic in nature?
This is because the metallic character of elements increases while traversing down a group, and radium is at the bottom of group 2 in the modern periodic table. The least metallic group 2 element is beryllium.
How metallic character varies in a group?
Metallic character increases down a group due to increase in atomic size. Thus, the outermost electrons are farther away from the nucleus and therefore can be lost easily. Hence, the electropositive character or metallic character increases.
How do you find the metallic character?
Metallic character can be measured by the ease of an element to give away its valence electrons. It depends on the valence electron’s i) effective nuclear charge (ENC) and ii) distance from the nucleus. Let’s see what’s the periodic trend of Metallic character.
Which element in Group 2 has the highest ionization energy?
And thus neon, with the greatest nuclear charge of the 2nd period, has the corresponding greatest ionization energy of the Period. Clearly, this property is shared by all the Noble Gases.
Which element in group 2 is most reactive?
The alkali metals are the most reactive metals. Group 2 consists of the alkaline Earth metals. They are very reactive but less so than the alkali metals.
What is the alkali metal in period 2?
Lithium is the first alkali metal in the periodic table, and the first metal of any kind in the periodic table. At standard temperature and pressure, lithium is a soft, silver-white, highly reactive metal.
Is Aluminium the most metallic?
Aluminum and phosphorus belong to the same period and we know that across the period metallic character increases from right to left. Aluminum is present on the left of phosphorus so it will be most metallic in nature.
Which is more metallic sodium or Aluminium Why?
We can see that sodium and aluminium are in the same period (3) and electrons in aluminium are added in the same shell as that of sodium. Thus, the effective nuclear charge is more on aluminium than sodium and thus aluminium has a smaller size. … This makes sodium more metallic than aluminium.
Which element is most metallic in 4th period?
The most metallic element in the fourth period is option 1-Ca ,2-K.
Which element is more metallic Ga or be?
Among the elements Ga, Ge, As, Se Be in the question. Be and Ga are expected to be most metallic. Out of Be and Ga, Ga is bigger in size and hence has a greater tendency to lose electrons than Be. Therefore, Ga is more metallic than Be.
Which is more metallic gallium or germanium?
But as we move down the group, the metallic character of the elements increases. … Now in germanium and gallium, gallium is more metallic because it belongs to group 13 and germanium belongs to group 14.
Which is more metallic beryllium or gallium?
Of course Gallium is more metallic , as Beryllium its an alkaline earth metal of group 2 and period II which has s-character which is more penetrating towards nucleus and so does not easily donate its electron, while Gallium is in 13th group in 3rd period it is a p-block element here the 3d shell is filled and it …