Why do successive ionization energies of an element increase

Sarah Rodriguez
Published Feb 16, 2026
Successive ionization energies increase in magnitude because the number of electrons, which cause repulsion, steadily decrease. … So, the amount of energy needed to remove electrons beyond the valence electrons is significantly greater than the energy of chemical reactions and bonding.
Why do successive ionization energies increase quizlet?
When do large increases in successive ionization energies occur? Because in doubly filled orbitals electrons repel each other. … It increases because the e are being added to the same shell which has the same distance from the nucleus and same shielding effect.
How do successive ionisation energies provide evidence for electron shells?
Each successive ionisation energy is bigger than the previous one for the same reason. Some of the increases are much bigger, however, and these big jumps gives us evidence for the main principle electron shells.
Why does ionization energy increase with each electron removed?
The ionization energy required for removal of electrons increases progressively as the atom loses electrons, because the positive charge on the nucleus of the atom does not change, and therefore, with each removal of an electron, the remainder are held more firmly.How successive ionization energies account for the existence of three main energy levels in the sodium atom?
The successive ionisation energies of Sodium show large increases in ionization energy when the 2nd and 10th electrons are removed. This shows that the 1st electron is further from the nucleus than the 2nd electron and same with the 9th and 10th electrons.
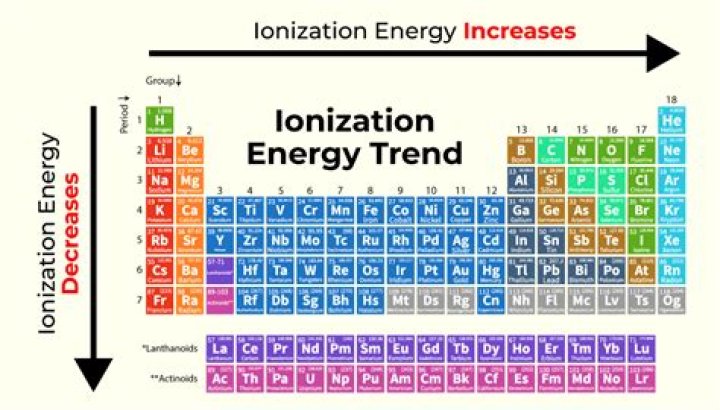
Why ionization energies increase you move from left to right across a period and decrease as you move down a group?
The ionization energy of the elements within a period generally increases from left to right. This is due to valence shell stability. The ionization energy of the elements within a group generally decreases from top to bottom. This is due to electron shielding.
What is successive ionization energy?
The successive ionization energy can be defined as the energy required for the removal of the second electron from the ion which suggests that 1 mole of gas with 1st ion having +1 charge further gives +2 charge atom by losing one more electron from its shell.
When comparing the successive ionization energies of an element an unusually big increase in ionization energy is seen when?
Transcribed image text: When comparing the successive ionization energies of an element, an unusually big increase in ionization energy is seen when the first valence electron is removed. the second valence electron is removed. the first core electron is removed. the eighth electron of is removed.Why does ionization energy increase across a period and decrease down a group?
In general, ionization energy increases across a period and decreases down a group. Across a period, effective nuclear charge increases as electron shielding remains constant. … The increased distance weakens the nuclear attraction to the outer-most electron, and is easier to remove (requires less energy).
Why the ionisation energy of every element is endothermic?The ionisation process involves removing an electron from an atom or ion. Since there is an attraction between that electron and nucleus, energy is required to overcome this attraction. Hence ionisation is an endothermic process.
Article first time published onWhy does sodium have the highest second ionisation energy?
Since the sodium is a positive ion it has a stronger positive charge so the electrons are now more strongly attracted to the sodium ion to remove an electron from the sodium ion it will require more energy.
How successive ionisation energy data is related to the electron configuration of an atom?
Electron configuration is also referred to as electronic configuration. Ionization energy, or ionisation energy, is the energy required to remove an electron from a gaseous atom or ion. … The greater the value of the ionization energy, the harder it is to remove an electron from the gaseous species.
How successive ionisation energy could determine the group of the element?
A specific element’s pattern of consecutive ionization energies provides insight to the electronic configuration of its atoms. Generally, ionization energy increases as more electrons are removed. Removing electrons from positively charged ions requires more energy.
What is second ionization energy trend?
Second ionization energy decreases as you go down the group. Third ionization energy decreases as you go down the group. For each element in the Group, the first ionization energy is less than the second ionization energy which is less than the third ionization energy.
Why is second ionisation enthalpy higher than first?
The first ionisation energy removes the electrons from a neutral atom while the second ionisation energy removes electrons from a positive atom. The electrons are tightly bound by the positive atom due to increased attraction force, therefore the second ionisation energy is higher than the first.
Where does the jump occurs for successive ionization energies?
jumps in ionization energy occur when we get to the shell below the valence shell, it’s a whole lot harder to remove an electron closer to the nucleus than it is farther away. So when we move to the shell below, those electrons are closer to the protons in the nucleus, harder to remove needs more energy.
How does ionization energy change when we move from left to right in a period in a periodic table?
Moving left to right within a period or upward within a group, the first ionization energy generally increases. As the atomic radius decreases, it becomes harder to remove an electron that is closer to a more positively charged nucleus.
Why does ionisation potential increases across a period?
Across a period from left to right, the ionisation energy increases. This is due to the increase in nuclear charge having a greater pull on the electrons and therefore more energy is required to remove electrons. Going down a group, the ionisation energy decreases.
What happens to the ionization energy when looking at atoms from left to right on the periodic table eg lithium to neon )?
Ionization energy refers to the amount of energy needed to remove an electron from an atom. Ionization energy decreases as we go down a group. Ionization energy increases from left to right across the periodic table.
Why does ionization energy decrease down a group quizlet?
Why does ionization energy decrease as you move down the periodic table? Because outer electron are further away from the nucleus as you go down a group, they feel less pull from the nucleus, so they are easier to remove. Electronegativity is the ability of an atom to attract electron in a chemical bond.
Does ionization energy increase decrease or remains the same as you go down group IIA explain why?
When we move down a group in the periodic table, more energy levels are added, and so valence electrons would become further and further away from the positive nucleus. … The less attraction between the electrons and the nucleus, the easier they are to remove—decreasing ionization energy.
Which of the following elements has the largest first ionization energy?
The first ionization energy varies in a predictable way across the periodic table. The ionization energy decreases from top to bottom in groups, and increases from left to right across a period. Thus, helium has the largest first ionization energy, while francium has one of the lowest.
Which one of these atoms has the largest effective nuclear charge?
For example, why is the second ionization energy higher than the first, and the third higher than the second? The same number of protons in the nucleus but less electrons hence the effective nuclear charge is higher for subsequent ionizations. Define electron affinity. The ability of an atom (or ion) to gain electrons.
Why do elements within a group have similar chemical properties?
In chemistry, a group is a vertical column in the periodic table of the chemical elements. … Each element within a group has similar physical or chemical properties because of its atom’s outermost electron shell (most chemical properties are dominated by the orbital location of the outermost electron).
Is ionization energy exothermic or endothermic Why?
Ionization energy is endothermic because it requires an energy input to occur.
Why is there a large increase in ionization energy when the 4th electron is removed from aluminum?
There is a significant increase in the twelfth ionization energy for aluminium because you are removing an electron from a closed shell.
Why is the second ionization energy of sodium about three times greater than the second ionization energy of magnesium?
~The second ionization energy of sodium is about three times greater than the second ionization energy of magnesium. ~Na+1 is already in the preferred form. Because of this, the second ionization energy of sodium is higher than normal. Mg+1 loses an electron to form s2 p6 .
Why the second ionisation energy of sodium is considerably higher than the second ionisation energy of magnesium?
For the 2nd IE of Na an electron is being removed from the 2p subshell. For the 2nd IE of Mg an electron is being removed from the 3s subshell.It requires more energy to remove an electron from a lower energy shell. The shielding on the electron is greater in Mg+.
Does ionization energies increase with an increase in the magnitude of the electron affinity?
Ionization energies increase with an increase in the magnitude of the electron affinity. Ionization energies decrease diagonally across the periodic table from He to Cs. Ionization energies depend on electron configuration.
How do successive ionisation energies provide evidence for electron shells?
Each successive ionisation energy is bigger than the previous one for the same reason. Some of the increases are much bigger, however, and these big jumps gives us evidence for the main principle electron shells.
How does ionisation energy provide evidence for sub shells?
Ionisation energy – the evidence for shells and sub-shells As electrons are negatively charged and protons in the nucleus are positively charged, there will be an attraction between them. The greater the pull of the nucleus, the harder it will be to pull an electron away from an atom.