Why does water evaporate in an open container

Sarah Rodriguez
Published Mar 22, 2026
evaporation, change of a liquid into vapor at any temperature below its boiling point. For example, water, when placed in a shallow open container exposed to air, gradually disappears, evaporating at a rate that depends on the amount of surface exposed, the humidity of the air, and the temperature.
Does water evaporate in an open container?
Now in an open container, the liquid water (or other liquid) will slowly evaporate, and the container will empty eventually. In a closed container, there is dynamic equilibrium between the vapour phase, and the liquid phase, and the rate of evaporation is equal to the rate of condensation.
Why does water evaporate when left uncovered?
At room temperature, there is evaporation (I wouldn’t call it excitation). This is because there are a few molecules of water which can manage to muster enough energy to escape from the large body of molecules and escape into air.
Why does liquid evaporate when it is kept in open?
When a molecule near the surface absorbs enough energy to overcome the vapor pressure, it will escape and enter the surrounding air as a gas. … Evaporation of water occurs when the surface of the liquid is exposed, allowing molecules to escape and form water vapor; this vapor can then rise up and form clouds.What happens to water when it is left in an open container for some time?
A puddle of water left undisturbed eventually disappears. The liquid molecules escape into the gas phase, becoming water vapor. Vaporization is the process in which a liquid is converted to a gas. Evaporation is the conversion of a liquid to its vapor below the boiling temperature of the liquid.
What happens to the water inside the small container?
2 Answers By Expert Tutors. Liquid water is always evaporating because statistically there are always molecules near the liquid-vapor surface which have enough energy to escape as gas molecules to become water vapor.
Is water the only liquid that can evaporate?
A: All liquids (and even solids) evaporate in the sense that some of their molecules or atoms fly off the surface into the nearby gas. If something blows those molecules away, then more will keep evaporating until the liquid is gone. If the whole thing is in a sealed box, however, an equilibrium is reached.
What happens when water evaporates at room temperature in an open container?
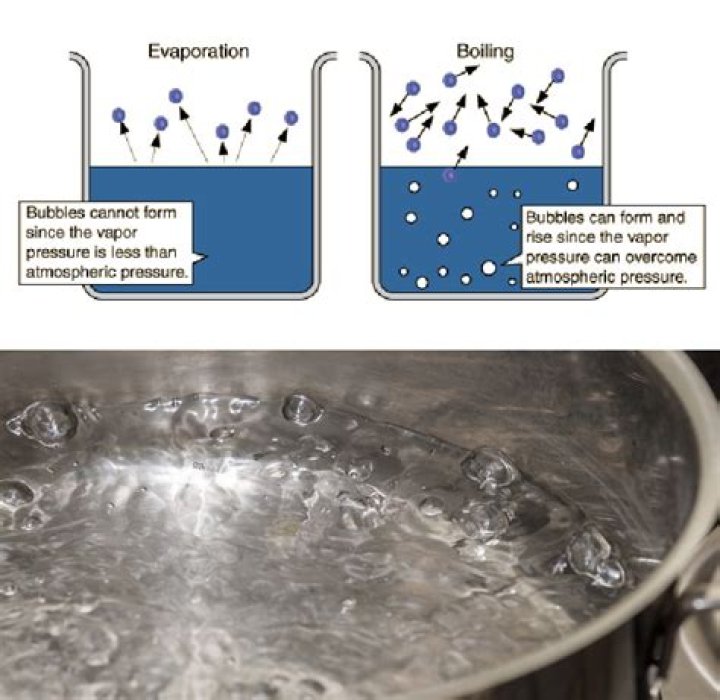
When liquid evaporates, individual molecules leave the liquid and become part of the air. In Figure 2, the dots represent molecules. The ones that leave the liquid and become part of the gas (air) are evaporating. The ones that go from gas to liquid are condensing.Where does liquid go during evaporation process?
Evaporation is the process by which water changes from a liquid to a gas or vapor. Evaporation is the primary pathway that water moves from the liquid state back into the water cycle as atmospheric water vapor.
Why does water evaporate in cold weather?Evaporation really depends on vapor pressure. … As the temperature of your pool’s water gets colder, the differential pressure between the water and the air increases. This speeds up the rate of evaporation. In addition, lack of humidity and wintertime dry air can also increase evaporation rates.
Article first time published onCan water evaporate below 100 degrees?
Liquid water is made up of molecules of H2O attracted to one another by intermolecular forces known as ‘hydrogen bonds’. These are relatively weak, and there are always some H20 molecules whizzing around with enough energy to break free of their neighbours, even at temperatures well below 100°C.
Does water evaporate without heat?
If you’re asking if water can evaporate without adding heat, the answer is yes: the heat source is the water itself. Like everything else, water has a certain equilibrium Vapor Pressure that depends on temperature.
What is disappearing water?
In the disappearing water trick, a magician makes water seem to disappear from inside a cup. … Essentially, you pour some water into a cup and wait a few moments. Then you turn the cup over, but no water pours out—the water has apparently vanished.
What made the water disappear?
In nature, the energy, or heat of the sun causes water to evaporate into its gaseous, or vapor, phase. Likewise, when we boil water over a burner we are causing it to change from a liquid to a gas. The process by which a substance changes from a liquid to a gas is called evaporation.
What will happen when you leave a closed container and an open container of water outside in the sunlight?
When you go back to observe the containers, the open container will have lost water. … Heat from the sun turns liquid water into its gas phase, water vapor. The water vapor then evaporates and is cools back into liquid water and eventually becomes part of a cloud.
What affects evaporation?
Liquids changes into vapour by the process of evaporation. The factors that affect the rate of evaporation of liquids are temperature, surface area, wind speed, and humidity.
Why do some liquids evaporate faster than others?
A substance that has a larger surface area will evaporate faster, as there are more surface molecules per unit of volume that are potentially able to escape. the higher the temperature of the substance the greater the kinetic energy of the molecules at its surface and therefore the faster the rate of their evaporation.
How do water molecules evaporate?
Evaporation happens when a liquid substance becomes a gas. When water is heated, it evaporates. The molecules move and vibrate so quickly that they escape into the atmosphere as molecules of water vapor. … Heat from the sun, or solar energy, powers the evaporation process.
What would happen to the water in both containers if the containers were closed?
Likely it will never boil. If the temperature in the closed system is the same everywhere, the pressure above the liquid will always be at or above the vapor pressure of the liquid. This will stop bubbles of vapor from forming inside the liquid. Hence there is no boiling.
Why is there water on the outside of my cup?
Condensation can also produce water droplets on the outside of soda cans or glasses of cold water. When warm air hits the cold surface, it reaches its dew point and condenses. This leaves droplets of water on the glass or can. When a pocket of air becomes full of water vapor, clouds form.
How do puddles evaporate?
The higher the temperature of the water at the surface, the more likely is it for these molecules to break free. The energy from the sun shining on it makes this godfather. They then turn into individual floating water molecules, aka vapour. Over time the entire puddle will evaporate that way.
How do you explain evaporation to a child?
Evaporation is a process where liquids change to a gas or vapor. Water changes to a vapor or steam from the energy created when molecules bounce into one another because they’re heated up. Sweat drying from our body is a great example of evaporation.
Why does water evaporate below its boiling point?
Evaporation can take place at temperatures below boiling point since the molecules in the liquid have different energies. … This process lowers the energy of the remaining molecules and is the source of cooling in evaporating liquids.
Why do puddles of rain water dry up?
Students will be able to explain that puddles dry up because tiny particles of water (water molecules) break away from the puddle and go into the air. … This process is known as the water cycle.
Does water evaporate in cold weather?
On cold days, water evaporates, but it evaporates more slowly than it would on a warmer day. Although water can evaporate at low temperatures, the rate of evaporation increases as the temperature increases.
How cold does it need to be for water to evaporate?
Water (liquid state) can evaporate at any temperature from 0 degrees to 100 degrees Celsius at Standard Pressure. The warmer the water, the faster it evaporates. Don’t confuse evaporation with sublimation. With sublimation, water can leave ice directly to the gaseous phase.
Does water evaporate without sunlight?
Yes. Even without the light, evaporation will take place when the partial pressure of water in the air is less than the vapour pressure of water at the ambient temperature.
Does water evaporate from your hair?
While you’re getting your 8 hours of beauty rest moisture is evaporating from your strands into the night air. That’s why hair can feel dry and straw-like after being slept on, uncovered, for hours. You can slow down the evaporation process by covering your hair at night, thereby keeping moisture locked in.
How do they do the milk illusion?
Milk is injected into the narrow space between the liner and the wall of the pitcher, making it appear full. As the pitcher is tipped, a shield on the lip of the pitcher creates a barrier and the milk flows through a cutout in the liner wall into the liner instead of pouring into the cone.
What happened to the water in glass?
Condensation on Glass When the hot air comes in contact with the cold glass, heat is transferred from the hot air to the cold glass. The loss of heat in the surrounding air causes the water vapor by the glass to lose energy. Once energy is lost, the water vapor condenses into liquid on the glass.
Where does the other half of the water went?
The right bar shows the breakdown of surface freshwater. Most of this water is locked up in ice, and another 20.9% is found in lakes. Rivers make up 0.49% of surface freshwater. Although rivers account for only a small amount of freshwater, this is where humans get a large portion of their water.