Why is HF a weak acid than HCl

Olivia Owen
Published May 08, 2026
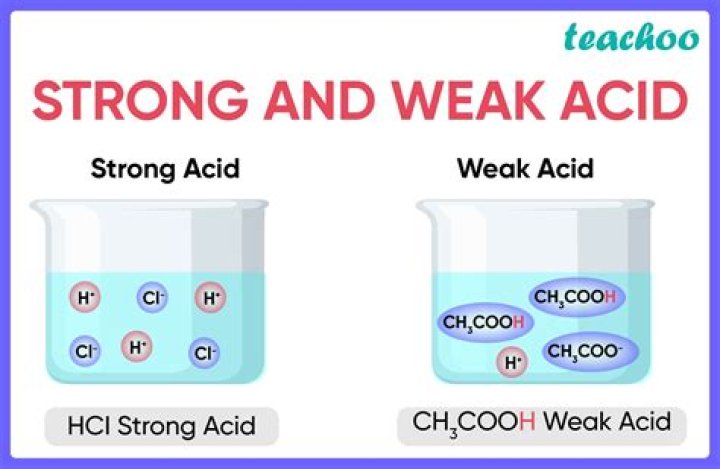
Why is HF a weaker acid than HCl ? … The hydrogen-chlorine bond in HCl is very weak, allowing it to completely dissociate in water (thus qualifying it as a strong acid). The hydrogen-fluorine bonding HF is relatively strong so it only partially dissociates in water, making it a weak acid.
Why is HCl a stronger acid than HF?
HCl is a stronger acid than HF because fluorine is more electronegative than chlorine.
Why HF is the weakest acid?
Hydrofluoric acid (HF) is chemically classified as a weak acid due to its limited ionic dissociation in H 2 O at 25°C [26]. In water at equilibrium, non-ionized molecules, HF, remain present and provides slowly H + and F − to form F − ·H 3 O + [26, 27].
Why is HF a weak acid as compared to HCl and HBr?
Fluorine has the shortest bond to the proton, which gives it the greatest bond dissociation enthalpy, so ultimately HF is weaker than HCl, which in turn is weaker than HBr, which is weaker than HI. This is attributable to the fact that the bond length gets larger as you go down the group (and thus weaker).Is hydrofluoric acid a weak acid?
This leads to protonation of very strong acids like hydrochloric, sulfuric, or nitric when using concentrated hydrofluoric acid solutions. Although hydrofluoric acid is regarded as a weak acid, it is very corrosive, even attacking glass when hydrated.
Why is HF a weak electrolyte?
HF, on the other hand will ionize in water (becoming H+ and F− ), but only to a small extent, because it is a weak acid. Therefore, the solution consists of few ions, and conducts very slightly. HF is the weak electrolyte.
Why is HF a weak acid but very reactive?
HF(aq) is a very reactive, weak acid. … The great attraction that fluorine has for most other atoms is the cause of HF(aq) being a weak acid. This high attraction of fluorine for most other atoms gives rise to a very strong H—F bond that is not broken when HF dissolves in water.
Is HF a strong or weak electrolyte?
Strong Electrolytesstrong acidsHCl, HBr, HI, HNO3, HClO3, HClO4, and H2SO4strong basesNaOH, KOH, LiOH, Ba(OH)2, and Ca(OH)2saltsNaCl, KBr, MgCl2, and many, many moreWeak Electrolytesweak acidsHF, HC2H3O2 (acetic acid), H2CO3 (carbonic acid), H3PO4 (phosphoric acid), and many moreIs HF a strong acid?
HCl, HBr, and HI are all strong acids, whereas HF is a weak acid. The acid strength increases as the experimental pKa values decrease in the following order: … Hydrochloric acid: Hydrochloric acid is a clear, colorless solution of hydrogen chloride (HCl) in water.
Why HF is the weakest acid and HI is the strongest?In HI iodine is less electronegative and it has large size. Therefore bonding between hydrogen and iodide is weaker. It will easily split in comparision of HF. Due to more liberation of H+ ions HI is stronger acid.
Article first time published onWhy HF is weak acid where as the binary acids of all others halogens are strong acids?
Because fluorine is at the top of the halogens, the F–ion is the smallest halide; therefore, its electrons are concentrated around its nucleus, and as a result, the H-F bond is relatively short. Shorter bonds are more stable, and thus the H-F bond is more difficult to break.
Is Hcooh a strong or weak acid?
Formic acid (HCOOH) is a weak acid. We know it is an acid because it contains the carboxyl (COOH) functional group, which makes it a carboxylic acid….
Is HL a weak acid?
There are only a few (7) strong acids, so many people choose to memorize them. All the other acids are weak. The strong acids are hydrochloric acid, nitric acid, sulfuric acid, hydrobromic acid, hydroiodic acid, perchloric acid, and chloric acid.
Which is more corrosive HCl or HF?
A certain percentage of the HF molecules will not disassociate. HF is therefore regarded as a weak acid. Interestingly, even though HCl stronger acid than HF, HF is by far the more dangerous of the two. The danger comes from the fluoride ion.
Why is hydrofluoric acid corrosive?
Fluorine forms VERY strong covalent bonds, and it forms them very readily. HF destroys you because very small molecules can penetrate deeply, and HF is small and fluorine can replace OH in most molecules and having replaced OH it stays put. Hydroxyl groups are key parts of the molecules of life.
Why is HF a weak acid class 9?
HF is a weak acid because its ability to dissociate to donate proton is low. It is not strong because it can dissolve glass. Some strong acids are not very corrosive, and some weak acids can be very corrosive.
How strong is hydrofluoric acid?
Hydrofluoric acid is extremely corrosive! Aqueous solutions dilute as 0.1 M will pH at approximately 1.0. Unlike other mineral acids, hydrofluoric acid will attack glass, concrete, rubber, quartz and alloys containing silica.
Is fluoride a weak base?
Fluoride is classified as a weak base since it only partially associates in solution, but concentrated fluoride is corrosive and can attack the skin. Fluoride is the simplest fluorine anion.
WHY IS F a weak base?
It is a weak base because F is a most electroneagtive elements which has more affection towards electron and it will never donates electron to other so it is a weak base.
Is HF a stronger acid than HI?
HI is a stronger acid than HF. The term “effective” is employed because the conversion of gaseous hydrogen atom to aqueous proton has been ignored in both analyses.
Why is HF more stable than HI?
Fluorine has the shortest bond to the proton, which gives it the greatest bond dissociation enthalpy, so ultimately HF is weaker than HCl, which in turn is weaker than HBr, which is weaker than HI. This is attributable to the fact that the bond length gets larger as you go down the group (and thus weaker).
Why HI is a stronger acid than HCl?
HI is stronger acid then HCl because dissociation energy of H–I bond is less than that of H–Cl.
Which is the weakest of these binary acids?
HOI is the weakest acid, because the electronegativity of Iodide is weaker therefore the acid bond strength is weaker.
What does HF mean in chemistry?
Hydrogen fluoride is a chemical compound that contains fluorine. It can exist as a colorless gas or as a fuming liquid, or it can be dissolved in water. When hydrogen fluoride is dissolved in water, it may be called hydrofluoric acid.
Is h2s04 a binary acid?
Binary acids, such as hydrochloric acid, HCl(aq). Oxyacids, such as sulfuric acid, H2 SO4 , and nitric acid, HNO3 . Organic acids (most often called carboxylic acids), such as acetic acid, HC2 H3 O2 . The names have the general form of hydro(root)ic acid, such as hydrochloric acid.
Why is Hcooh a weak acid?
HCOOH is a weak acid because it contains fewer hydrogen ions in the final aqueous solution as it only dissociates partially or not 100% ionized when dissolved in water. The conjugate base of HCOOH is the Formate ion (HCOO–).
Can weak acids Protonate?
Strong acids such as HCl will give up protons even at very low pH (hence low pKa) and weak acids will only give up protons if the pH is very high (i.e. the free proton concentration is very low).
Why is HPO4 a weak acid?
Salts containing the anion H2PO4‾ are weakly acidic. The tendency of this ion to dissociate is greater than its tendency to hydrolyse, that is, its Ka2, is larger than its Kb. … Salts containing the anion HPO4‾ are weakly basic. The tendency of this ion to hydrolyse is greater than its tendency to dissociate.
Is h2s04 a weak acid?
HSO4– is a weak acid and stable conjugate base of H2SO4, hence, it will not reform with H+ to forms H2SO4 again in an aqueous solution. In the second reaction, HSO4– as a weak acid that also partially dissociate and release the one proton and forms SO42-.
Is Ba2+ a weak acid?
The parent acids are so strong in water that the conjugate bases are exceedingly weak. Cations of the group 1A metals (Li+, Na+, K+, Rb+, Cs+) and the group 2A metals (Ca2+, Sr2+, Ba2+) do not react with water and are nonacids.
What makes a weak acid?
A weak acid is an acid that partially dissociates into its ions in an aqueous solution or water. In contrast, a strong acid fully dissociates into its ions in water. … At the same concentration, weak acids have a higher pH value than strong acids.