Why is Isoborneol favored over borneol

Isabella Wilson
Published Mar 16, 2026
The peak differences show that isoborneol was preferred over borneol, due to the steric strain caused by the two geminal methyl groups on one side of the cyclohexane. Since there is still a camphor peak on the gas chromatography spectra, the reduction reaction did not go to completion.
Is isoborneol more stable than borneol?
Though borneol is the more stable product, the energy requirements to form isoborneol are lower because the borohydride is adding to the less sterically hindered point on the carbonyl carbon.
What is the difference between borneol and isoborneol?
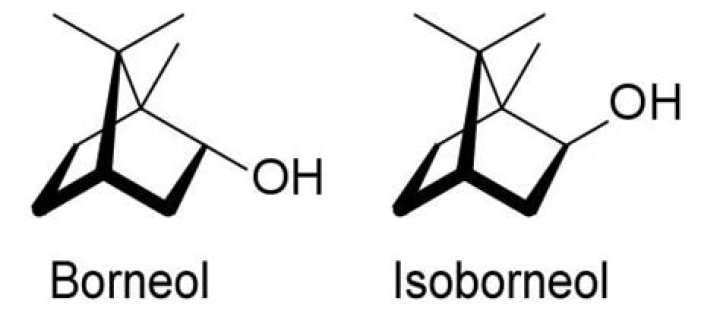
An alcohol (borneol) is being oxidized into a ketone (camphor). A subsequent reduction takes us back to another alcohol (isoborneol), which is an isomeric form of the original.
Why is isoborneol the major product of reaction?
The Exo product is the major product because endo is favorable (less hindered). The driving force is the formation of very strong B-O-bond (ΔH=523 kJ/mol) in the tetraalkyl borate which are significantly stronger than π-bonds in the carbonyl (ΔH=380 kJ/mol).Is isoborneol the endo product?
During the reduction of camphor, the reducing agent can approach the carbonyl face with a one carbon bridge (termed exo attack) or the face with a two carbon bridge (termed endo). The two product stereoisomers are called borneol (from exo attack) and isoborneol (from endo attack).
What is the melting point of Isoborneol?
DL-Isoborneol Properties Melting point: 212-214 °C (subl.)(
Is Isoborneol or borneol favored?
The peak differences show that isoborneol was preferred over borneol, due to the steric strain caused by the two geminal methyl groups on one side of the cyclohexane.
Is borneol soluble in water?
3.2Experimental Properties Borneol appears as a white colored lump-solid with a sharp camphor-like odor. Burns readily. Slightly denser than water and insoluble in water.How many chiral centers does borneol have?
Notice that there are actually 3 chiral centers in the (1S)-[endo]-(1)-borneol substrate.
What functional group is the starting material camphor?Camphor has a ketone functional group.
Article first time published onDoes isoborneol have hydrogen bonding?
Property NameProperty ValueReferenceHydrogen Bond Acceptor Count1Computed by Cactvs 3.4.8.18 (PubChem release 2021.05.07)
Which statement would correctly describe isoborneol?
Which statement would correctly describe isoborneol? It is a bridged bicyclic alcohol. How might the IR of isoborneol differ from that of camphor? Isoborneol should show an O-H stretch at 3600-3200 cm-1.
Is isoborneol a primary alcohol?
Isoborneol is one of its corresponding secondary alcohols such that this carbon atom has a bond with hydrogen and a bond with the hydroxide ion (H-C-OH). … Chemically, a ketone (camphor) may be converted into one of its secondary alcohols (isoborneol) with a reducing agent (sodium borohydride).
How do you separate Isoborneol and borneol?
Borneol (I) and isoborneol (II) in synthetic Bingpian are separated by means of dry-column chromatography (DCC).
How will you distinguish borneol Isoborneol and camphor by NMR?
In NMR, the CH(OH) peak differed by 0.5 ppm for borneol and isoborneol. Camphor and the remaining borneol/isoborneol peaks couldn’t be distinguished by NMR. TLC is not an ideal technique to monitor the borneol oxidation. … Only the reactant borneol can be seen on the TLC, not the product camphor.
Does borneol sublime?
Industrially, it can be obtained from α-pinene by two rearrangements reactions. The hydrolysis of the isobornyl acetate leads to borneol that is oxidized to form racemic camphor. … The crude camphor is purified by sublimation.
Why is reduction of camphor stereoselective?
In principle, the reduction of camphor can give two diastereomeric alcohols, corresponding to reaction of borohydride at the two faces of the C=O. … Since “endo-attack” of borohydride avoids this steric interaction, we might predict that it should proceed faster than exo-attack, leading to stereoselective reduction.
How is Isoborneol flakes made?
Borneol can be synthesized by reduction of camphor by the Meerwein–Ponndorf–Verley reduction (a reversible process). Reduction of camphor with sodium borohydride (fast and irreversible) gives instead the isomer isoborneol as the kinetically controlled reaction product.
How many diastereomers does borneol have?
Chemically synthesized borneol contains four stereoisomers, (+)-isoborneol, (−)-isoborneol, (−)-borneol, and (+)-borneol.
Are diastereomers Nonsuperimposable?
Diastereomers are the stereomer compounds with molecules that are not mirrored images of one another and that are not superimposable. They are non-superimposable mirror images of each other.
What is the major product of the reduction of camphor?
Camphor and its reduction products, borneol and isoborneol, come from a bicyclic family called terpenes.
Is Isoborneol toxic?
May be harmful by inhalation, ingestion, or skin absorption. May cause eye, skin, or respiratory system irritation.
What is borneol good for?
A core element in Chinese herbal medicine, borneol is used to facilitate digestion, improve circulation, and ease pain brought on by rheumatic diseases. Chinese herbalists also use the bitter tasting terpene to treat bronchitis, coughs, and colds while recognizing its stress-relieving qualities.
Is borneol safe?
* Borneol can irritate the nose and throat. * Exposure to Borneol can cause headache, nausea and vomiting, and can make you feel dizzy, lightheaded and pass out. * Higher exposure can cause restlessness, difficulty concentrating, irritability and seizures. * Borneol may cause a skin allergy.
What is the reason borneol and camphor are not UV active?
Therefore, borneol is not UV active. … The reason is that borneol has very similar chemical and physical properties (including molecular weight) as camphor. Any unreacted borneol would likely mix with the crude camphor and cause the % crude yield to remain unchanged.
Why camphor is volatile?
It is volatile in nature because it changes directly to gaseous state from solid state. Camphor is waxy, flammable and white.
What category is camphor?
Camphor belongs to a group of organic compounds defined as terpenoid ketones. … The structure and the reactions peculiar to it were important problems of 19th-century organic chemistry. The pure compound is a white, waxy solid that melts at about 178°–179° C (352°–354° F).
What is the density of Isoborneol in g ml?
Density:1.0±0.1 g/cm3Boiling Point:212.0±0.0 °C at 760 mmHgVapour Pressure:0.0±0.9 mmHg at 25°CEnthalpy of Vaporization:52.1±6.0 kJ/molFlash Point:80.7±10.9 °C
What intermolecular forces are present in biphenyl?
Biphenyl and Ligroin both have London disperson forces due to nonpolar C-C and C-H bonds which means biphenyl dissolves in ligroin very well.
Is methanol volatile?
Methanol appears as a colorless fairly volatile liquid with a faintly sweet pungent odor like that of ethyl alcohol. Completely mixes with water. … It is an alkyl alcohol, a one-carbon compound, a volatile organic compound and a primary alcohol. It is a conjugate acid of a methoxide.
How do you eliminate excess HOCl at the end of the reaction in the camphor experiment?
How do you eliminate excess HOCl at the end of the reaction in the camphor experiment? Add sodium bisulfite as a reducing agent.



