Why is it called bomb calorimeter

Sarah Rodriguez
Published Apr 17, 2026
Gases at high pressures are frequently used, hence the name. In 1878, Paul Vieille (1854–1934) developed the first bomb calorimeter which was used for measuring heats of explosion at the French service of explosives in Paris. However, this bomb was attributed by many authors to M. Berthelot (1827–1907).
Why is bomb calorimeter called bomb?
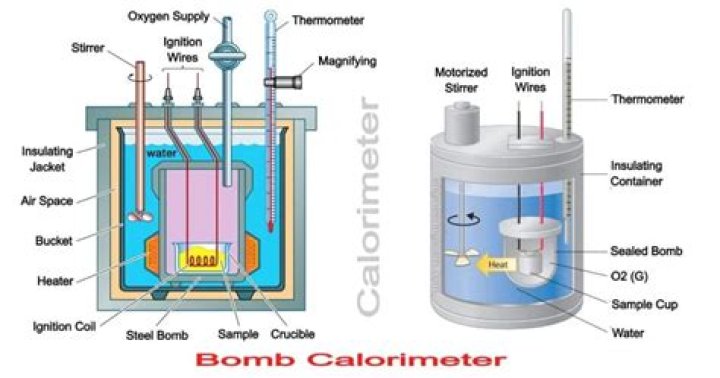
Bomb calorimeter consists of a strong steel vessel ( called bomb)which can stand high pressure when the substance is burnt in it. Hence, it is called bomb calorimeter.
What is the difference between a calorimeter and a bomb calorimeter?
A coffee cup calorimeter is great for measuring heat flow in a chemical solution, but it can’t be used for reactions, which involve gases since they would escape from the cup. … A bomb calorimeter is used to measure heat flow for solids with low to high temperature reactions.
What is meant by bomb in calorimeter?
noun. chem a device for determining heats of combustion by igniting a sample in a high pressure of oxygen in a sealed vessel and measuring the resulting rise in temperature: used for measuring the calorific value of foods.Why is it called a calorimeter?
The first ice calorimeters were built based on Joseph Black’s concept of latent heat, introduced in 1761. Antoine Lavoisier coined the term calorimeter in 1780 to describe the apparatus he used to measure heat from guinea pig respiration used to melt snow.
Who invented bomb calorimeter?
The next milestone takes us to Pierre Eugene Berthelot, French chemist, who built the first modern bomb calorimeter in the 1870-s. He is also credited with inventing the concept of endothermic and exothermic reactions [6].
What is the function of a bomb calorimeter quizlet?
A bomb calorimeter is an instrument that measures the heat energy released when food is burned, thus providing an estimate of the potential energy of the foods.
What is bomb calorimeter Slideshare?
Bomb Calorimeter • Bomb calorimeter is used to measure the changes in a system’s internal energy due to a reaction. • The basic principle is a chemical reaction heats a quantity of water in an insulated container. • The reaction takes place inside a sealed container, called as bomb.What is bomb calorimeter in chemistry class 11?
The bomb calorimeter consists of strong steel vessel which can withstand high pressure. It is surrounded by bigger vessel which contains water and is insulated and a thermometer and stirrer is suspended in it. The procedure consists of two steps: … Oxygen gas is introduced that is under high pressure in the bomb.
Is bomb calorimeter adiabatic?A bomb calorimeter is only approximately adiabatic. In reality, there is a small heat leak through the dewar (qcalorimeter ¹ 0) and the stirrer does work on the calorimeter (wcalorimeter ¹ 0).
Article first time published onWhat is the main characteristic of a calorimeter in a bomb calorimeter experiment?
The main characteristic of a bomb calorimeter is that it has a constant volume. It is essential because it can directly determine the enthalpy change of a sample by measuring the initial and final temperatures of the surrounding water.
Why is bomb calorimeter constant volume?
In a constant volume calorimeter, the system is sealed or isolated from its surroundings, which accounts for why its volume is fixed and there is no volume-pressure work done. A bomb calorimeter structure consists of the following: Steel bomb which contains the reactants. Water bath in which the bomb is submerged.
Does bomb calorimeter measure enthalpy?
The enthalpy changes that accompany combustion reactions are therefore measured using a constant-volume calorimeter, such as the bomb calorimeter(A device used to measure energy changes in chemical processes.
What principle do bomb calorimeters operate on?
Bomb calorimeters have to withstand the large pressure within the calorimeter as the reaction is being measured. Electrical energy is used to ignite the fuel; as the fuel is burning, it will heat up the surrounding air, which expands and escapes through a tube that leads the air out of the calorimeter.
What is the use of a bomb calorimeter Mcq?
Explanation: Bomb calorimeter can be used to determine the calorific value of both solid and liquid fuels.
What is the role of air jacket in bomb calorimeter?
The cooling/heating jacket controls the temperature of the process. Heat is measured by monitoring the heat gained or lost by the heat transfer fluid.
What does a bomb calorimeter measure nutrition quizlet?
What does a bomb calorimeter measure? The energy value of food.
Which of the following accurately describes fat cell development?
Of these foods, which is the best source of zinc? Osteocalcin ________. The DRI suggests a diet that provides ________ of the daily energy intake from fat. The body’s primary source of glucose during physical activity is _______.
What is the ultimate reason for the development of obesity?
Food and Activity. People gain weight when they eat more calories than they burn through activity. This imbalance is the greatest contributor to weight gain.
What is the meaning of calorimeter?
Definition of calorimeter : an apparatus for measuring quantities of absorbed or emitted heat or for determining specific heats.
How do I verify Hess law?
Finally, to verify Hess’s Law, you will use the H’s you measured for the first two reactions to calculate H for the third reaction. Compare it to what you measured for the third reaction by calculating the Percent Error.
What are the uses of bomb calorimeter?
Bomb calorimeters are devices used to determine the heat of combustion of a chemical reaction. The information gathered from a bomb calorimeter during a chemical reaction tells scientists whether certain products are safe for use and the quality level of each product being tested.
What are the two types of calorimeters?
There are two commonly used types of calorimeters: coffee cup calorimeters and bomb calorimeters.
What is the principle of calorimetry?
The principle of calorimetry (or principle of mixtures) states that for an insulated system, heat energy lost by the hot body is equal to the heat energy gained by the cold body. Note: Heat transfer occurs until both the bodies attain the same temperature(t).
Why is a bomb calorimeter surrounded by water?
One type in widespread use, called a bomb calorimeter, basically consists of an enclosure in which the reaction takes place, surrounded by a liquid, such as water, that absorbs the heat of the reaction and thus increases in temperature.
Why do you need to purge the nitrogen out of the bomb calorimeter?
The purging of nitrogen from the bomb interior eliminates corrections due to the heat of combustion of N2. … The mass of the water and bucket must be reproduced within 0.1% for all future combustion runs. Position the bucket inside the calorimeter jacket.
Which of the following process occurs in bomb calorimeter?
A bomb calorimeter is used to measure, under controlled conditions, the heat emitted by a sample burned under an oxygen atmosphere in a closed vessel (bomb) surrounded by water. The consequence of the calculation is called the amount of combustion, calorification, or BTU.
Does a bomb calorimeter have constant pressure?
Constant-Pressure Calorimetry In contrast, a bomb calorimeter ‘s volume is constant, so there is no pressure-volume work and the heat measured relates to the change in internal energy (ΔU=qV Δ U = q V ).
What is adiabatic calorimeter?
In principle, an adiabatic calorimeter is one in which heat is confined to the calorimeter usually by surrounding it with an adiabatic shield maintained at the temperature of the calorimeter. … The heat exchange during an experiment is commonly accounted for by the use of measurements at other times.