Do all proteins need chaperones

Isabella Wilson
Published Mar 13, 2026
Although most newly synthesized proteins can fold in absence of chaperones, a minority strictly requires them for the same. … Some chaperones can assist in protein degradation, leading proteins to protease systems, such as the ubiquitin-proteasome system in eukaryotes.
Why are chaperone proteins needed?
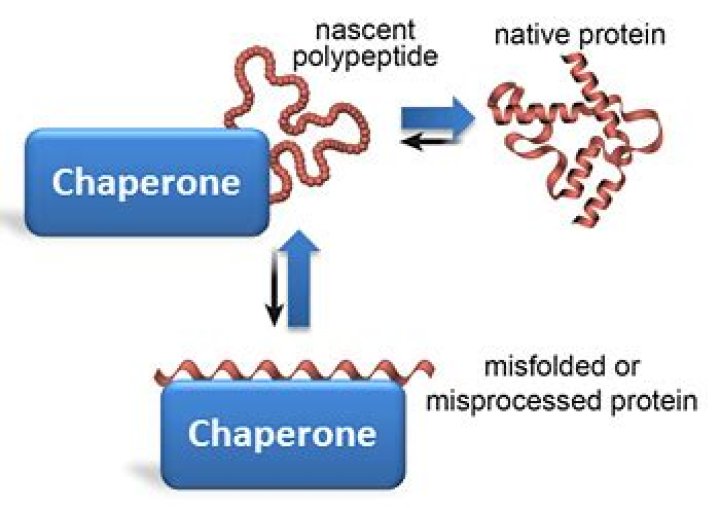
Chaperones are proteins that guide proteins along the proper pathways for folding. They protect proteins when they are in the process of folding, shielding them from other proteins that might bind and hinder the process. … Heat, in general, destabilizes proteins and makes misfolding more common.
Are all heat shock proteins chaperones?
Heat shock proteins (HSPs) are a group of proteins that function to reverse or inhibit denaturation or unfolding of cellular proteins in response to stress or high temperature. Traditionally, HSPs have also been known as molecular chaperones because of their physiological and protective roles in cells.
Are chaperones required for protein folding?
Molecular chaperones are essential to protein folding and can prevent protein aggregation by binding to non-specific proteins.What is the difference between a chaperone and a chaperonin?
The key difference between chaperons and chaperonins is that the chaperones perform a wide array of functions including folding and degradation of the protein, aiding in protein assembly, etc., whereas the key function of chaperonins is to assist in the folding of large protein molecules.
What type of protein is heat shock protein?
Heat shock proteins (Hsps) are a large family of evolutionarily conserved molecular chaperones with pivotal roles in cell survival and development. Hsps can be broadly classified into two families based on comparable molecular mass.
Why are chaperones called heat shock proteins?
Heat shock proteins (HSP) are a family of proteins that are produced by cells in response to exposure to stressful conditions. … Many members of this group perform chaperone functions by stabilizing new proteins to ensure correct folding or by helping to refold proteins that were damaged by the cell stress.
Do Chaperonins require ATP?
Chaperonins undergo large conformational changes during a folding reaction as a function of the enzymatic hydrolysis of ATP as well as binding of substrate proteins and cochaperonins, such as GroES.How do you activate cold shock proteins?
Cold showers. While they will not give you the benefits of more extreme full-body cold exposure, cold showers can ease you into cryotherapy and create mild cold shock protein activation. Ice baths. With ice baths, you have two options: longer and slightly cold, or shorter and very cold.
Where are molecular chaperones found?Chaperonins are characterized by a stacked double-ring structure and are found in prokaryotes, in the cytosol of eukaryotes, and in mitochondria. Other types of chaperones are involved in transport across membranes, for example membranes of the mitochondria and endoplasmic reticulum (ER) in eukaryotes.
Article first time published onDo chaperones require ATP?
During and after protein translation, molecular chaperones require ATP hydrolysis to favor the native folding of their substrates and, under stress, to avoid aggregation and revert misfolding.
What important function do molecular chaperones perform?
Molecular chaperones, including Hsp60s, Hsp70s, Hsp90s and sHsps, assist in the folding of unfolded and misfolded polypeptides by stabilization of folding intermediates and prevention of protein misfolding and aggregation. Several chaperones also function to reactivate aggregated proteins.
How might a cell protect itself when it is initially exposed to high heat?
Increased synthesis of heat shock proteins (hsp) occurs in prokaryotic and eukaryotic cells when they are exposed to stress. By increasing their hsp content, cells protect themselves from lethal assaults, primarily because hsp interfere with the uncontrolled protein unfolding that occurs under stress.
Are chaperone proteins enzymes?
Chaperones and foldases are two groups of accessory proteins which assist maturation of nascent peptides into functional proteins in cells. … It is suggested that the combination of chaperone and enzyme activities in one molecule is the result of evolution to increase molecular efficiency.
Do all organisms have heat shock proteins?
Heat-shock proteins (HSPs), or stress proteins, are highly conserved and present in all organisms and in all cells of all organisms.
What is the function of heat shock protein?
Heat shock proteins (HSPs) are molecular chaperones involved in a variety of life activities. HSPs function in the refolding of misfolded proteins, thereby contributing to the maintenance of cellular homeostasis.
Which one of the following is the main function of many proteins?
Protein has many roles in your body. It helps repair and build your body’s tissues, allows metabolic reactions to take place and coordinates bodily functions. In addition to providing your body with a structural framework, proteins also maintain proper pH and fluid balance.
What is a cold protein?
Cold shock proteins are multifunctional RNA/DNA binding proteins, characterized by the presence of one or more cold shock domains. … Biological activities range from the regulation of transcription, splicing and translation, to the orchestration of exosomal RNA content.
What are heat and cold shock proteins?
As one might expect, “cold shock” proteins appear when the body experiences a sudden decrease in its core temperature. Similarly, “heat shock” proteins go to work when the body enters a state of hyperthermia or elevated temperature.
What do RNA binding proteins do?
RNA-binding proteins (RBPs) have important functions in the regulation of gene expression. RBPs play key roles in post-transcriptional processes in all eukaryotes, such as splicing regulation, mRNA transport and modulation of mRNA translation and decay.
Are chaperonins lipids?
Lipids are hydrophobic and, consequently, they require chaperones, namely proteins that bind lipids, for their trafficking in the aqueous environment of cells, cellular compartments, and extracellular space typical of living organisms.
How do chaperonins fold proteins?
Chaperonins form a double ring structure stacked back-to-back, and assist protein folding in the central cavities (Fig. 1A). The class of chaperonins are subdivided into two groups.
How chaperonins may assist in proper folding of proteins?
Explain how chaperonins may assist in proper folding of proteins. The chaperonins keep the new polypeptide segregated from disruptive chemical conditions in the cytoplasmic environment while the protein folds spontaneously.
Which molecule acts as molecular chaperones to assist the folding of proteins?
Lipids are the molecule acts as molecular chaperones to assist the folding of proteins.
Do chaperones lower activation energy?
Water and chaperones may not only decrease the activation energy (the saddles) between the energy minima but may also transiently change the x–y plane of the landscape and the absolute value of the energy minima (the z axis).
What is the function of chaperone proteins quizlet?
Terms in this set (15) In molecular biology, molecular chaperones are proteins that assist the covalent folding or unfolding and the assembly or disassembly of other macromolecular structures.
What bonds hold proteins together?
Within a protein, multiple amino acids are linked together by peptide bonds, thereby forming a long chain. Peptide bonds are formed by a biochemical reaction that extracts a water molecule as it joins the amino group of one amino acid to the carboxyl group of a neighboring amino acid.
How the molecular chaperones plays role in the modification of the nascent proteins?
Rather, chaperones catalyze protein folding by assisting the self-assembly process. They appear to function by binding to and stabilizing unfolded or partially folded polypeptides that are intermediates along the pathway leading to the final correctly folded state.
Do proteins regulate cell processes?
Proteins are polymers of molecules called amino acids. Proteins perform many varied functions, such as controlling the rate of reactions and regulating cell processes, forming cellular structures, transporting substances into or out of cells, and helping to fight disease.
Do proteins denature at high temperatures?
The melting temperature varies for different proteins, but temperatures above 41°C (105.8°F) will break the interactions in many proteins and denature them. … Factors other than heat can also denature proteins. Changes in pH affect the chemistry of amino acid residues and can lead to denaturation.
What could happen to a person's proteins if their internal temperatures got too high?
This level of heat can threaten the functions of proteins, whose regular functioning depends on the body’s normal temperature. Severely high fever can cause seizures, confusion, throbbing headaches, unusual sensitivity towards bright light and sound, difficulty in breathing etc.