Does CBr4 have weaker intermolecular forces than CCl4

Lily Fisher
Published Feb 13, 2026
Stronger dispersion forces would occur between CBr4 molecules because it has a higher molecular weight than CCl4. Therefore, CCl4 would be more volatile, which results in a higher vapor pressure (when compared at the same temperature) and a lower boiling point than that of CBr4.
Does CBr4 have stronger intermolecular forces?
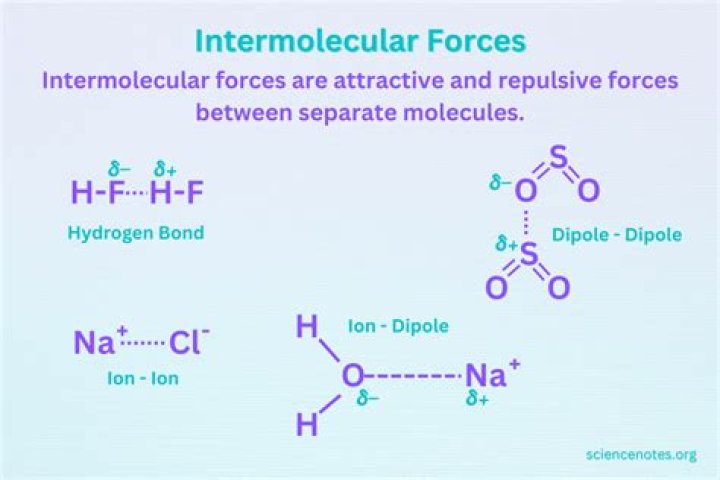
CCl4 C C l 4 and CBr4 C B r 4 are both nonpolar due to their highly symmetrical tetrahedral geometry. This means that only London dispersion forces will be the dominant forces that hold their molecules together and the major forces that must be overcome to bring the boiling point of the two compounds.
Why does CCl4 have stronger intermolecular forces?
Intermolecular forces in CCl4 The C-Cl bonds are polar but, because of the tetrahedral symmetry, the bond dipoles cancel each other. Thus, CCl4 is a nonpolar molecule, and its strongest intermolecular forces are London dispersion forces.
Does CCl4 have weak intermolecular forces?
However, CCl4 is nonpolar and thus has weak intermolecular forces of attraction. Water is polar and contains strong hydrogen bonds between molecules.Is CCl4 strongest intermolecular force?
CCl4 is a nonpolar molecule. Its strongest intermolecular forces are London dispersion forces.
What intermolecular forces are present between CBr4 molecules?
The main intermolecular force between two non-polar molecules (such as CBr4) are van der Waals’ forces which result from temporary dipoles formed as electron distributions become unbalanced. The extent of these interactions increases as more electrons are present (as in heavier molecules with larger atoms).
What intermolecular forces does CBr4 have?
QuestionAnswerWhat types of intermolecular forces exist between Br2 and CCl4?London dispersionWhat is the intermolecular force in CBr4?London dispersionOf the following substances, Kr, CH4, CO2, or H2O, which has the highest boiling point?H2O
Does CCl4 have dipole dipole forces?
Nonpolar molecules experience only induced dipole (dispersion or London) forces, and of the examples above, only CCl4 (l) and Br2 (l) are nonpolar.Does CBr4 have dipole dipole forces?
Next, we might be tempted to say that CBr4 also has dipole-dipole forces, but we have to consider the shape of the molecule. … You must have dispersion forces before you can have dipole-dipole forces, and you must have both dispersion and dipole-dipole forces before you can have hydrogen bonding.
Does CCl4 have stronger intermolecular forces than CH4?London dispersal forces are generally stronger when there are more electrons to form spontaneous dipoles, and hence often correlate with molecular weight. So CH4 would have the weakest forces and the lowest boiling point, CCl4 the highest.
Article first time published onWhich has stronger intermolecular forces HCl or CCl4?
The response earned 1 point in part (d)(ii) because the student correctly states that CCl4 has stronger intermolecular forces than HCl because CCl4 condenses at a higher temperature and has a higher boiling point.
What is the strongest intermolecular force to be overcome when dichloromethane?
Factor Affecting Boiling Point: The strongest among them is hydrogen bonding, and the weakest are London dispersion forces.
Does CCl4 or HCl have stronger intermolecular forces?
The dispersion forces between CCl4 molecules are stronger than the dipole forces between molecules of HCl since CCl4 is much larger.
Which has the strongest intermolecular force CCl4 or Cl2?
Cl2 and CCl4 are non polar and do not have a dipole moment. The strongest intermolecular force is dispersion forces.
Which intermolecular force is the strongest?
Dipole-dipole interactions are the strongest intermolecular force of attraction.
What type of intermolecular forces would you expect to be the predominant strongest forces in a sample of Xe?
SubstanceXeMolar Mass (g/mol)131Melting Point (°C)−111.8Boiling Point (°C)−108.1
Is CBr4 ionic or covalent?
The given compound carbon tetrabromide is a covalent compound. Its formula is CBr4 C B r 4 . The bond formed in carbon tetrabromide is covalent since the constituent atoms, carbon and bromine, are non-metals.
What intermolecular forces are present in C3H8?
Exhibits dipole and London dispersion forces but no hydrogen bonding since it has no H covalently bonded to the O. Propane, C3H8, has 3(4) + 8(1) = 20 valence electrons. Propane only has relatively nonpolar bonds, so it is nonpolar. Propane exhibits only London dispersion forces.
What intermolecular forces are present in ch3cooh?
In acetic acid (CH3COOH), hydrogen bonding, dipole-dipole interactions and dispersion force are present whereas in carbon tetrachloride (CCl4) only dispersion non-polar forces are present.
What types of intermolecular forces exist between Cl2 and CCl4?
Because both Cl2 and CCl4 are nonpolar and have no other special identifying characteristics, the only intermolecular forces between the two molecules are London Dispersion Forces.
Does CBR have a permanent dipole?
Although the carbon-iodine bond doesn’t have a permanent dipole, the bond is very easily polarised by anything approaching it. Imagine a negative ion approaching the bond from the far side of the carbon atom: The fairly small polarity of the carbon-bromine bond will be increased by the same effect.
Is CBr4 polar or nonpolar?
The CBr4 molecule is non-polar. The CH3Br molecule is polar. Both CBr4 and CH3Br have four regions of electrons around the central carbon atom. These are all bonding electron regions (clouds) so the shape of both molecules is tetrahedral.
Does CH2O have dipole dipole interactions?
The CH2O molecules can form London forces and dipole/dipole forces.
Is CH2O dipole dipole?
Due to their different three-dimensional structures, some molecules with polar bonds have a net dipole moment (HCl, CH2O, NH3, and CHCl3), indicated in blue, whereas others do not because the bond dipole moments cancel (BCl3, CCl4, PF5, and SF6).
Does CH3 2O have a dipole moment?
Yes, (CH3)2O ( C H 3 ) 2 O has a dipole moment. There is enough difference in electronegativity between oxygen and carbon (net 1.0) In looking at…
Why does CCl4 have no overall dipole?
– Carbon tetrachloride has the structure as shown below: – Here, the four bonds are the symmetrical ones and are extended in all directions and therefore, the dipole moments of each chlorine atom cancel each other which makes them non polar in nature and thus have zero dipole moment.
Is CCl4 polar or nonpolar?
CCl4 that is carbon tetrachloride is nonpolar because all the four bonds are symmetrical, and they are they extended in all the directions. This makes it easy for the dipole moments in each direction to cancel out.
What intermolecular forces does CH4 have?
The only intermolecular forces in methane are London dispersion forces. The major intermolecular forces would be dipole-dipole forces and London dispersion forces. The electronegativities of C and H are so close that C-H bonds are nonpolar.
When carbon tetrachloride CCl4 boils What changes in forces and energy occur?
London dispersion forces and carbon tetrachloride are stronger than they are in nothing and the stronger inter molecular forces result in a higher boiling point. It takes more energy to break those forces apart so that it can change from a liquid to a gas, so that gives you a higher boiling point.
Which of the following is the weakest intermolecular force?
London Dispersion Forces. The London dispersion force is the weakest intermolecular force. The London dispersion force is a temporary attractive force that results when the electrons in two adjacent atoms occupy positions that make the atoms form temporary dipoles.
Which liquid has the weakest intermolecular forces?
- Oil- Only London Dispersion Forces (the weakest intermolecular force)
- Water- London Dispersion, Dipole-Dipole, and Hydrogen Bonding.