Does the water molecule have single bonds or double bonds

Andrew Campbell
Published Feb 25, 2026
We have two hydrogens which can only form single bonds. Thus, in water, oxygen is singly bonded to two hydrogen atoms. In case of carbon dioxide, i.e. CO2, carbon is the central atom and there are two oxygen atoms. Carbon can form four bonds while oxygen can form a maximum of double bond.
Is water single or double bond?
We have two hydrogens which can only form single bonds. Thus, in water, oxygen is singly bonded to two hydrogen atoms. In case of carbon dioxide, i.e. CO2, carbon is the central atom and there are two oxygen atoms. Carbon can form four bonds while oxygen can form a maximum of double bond.
What type of bond does a water molecule have?
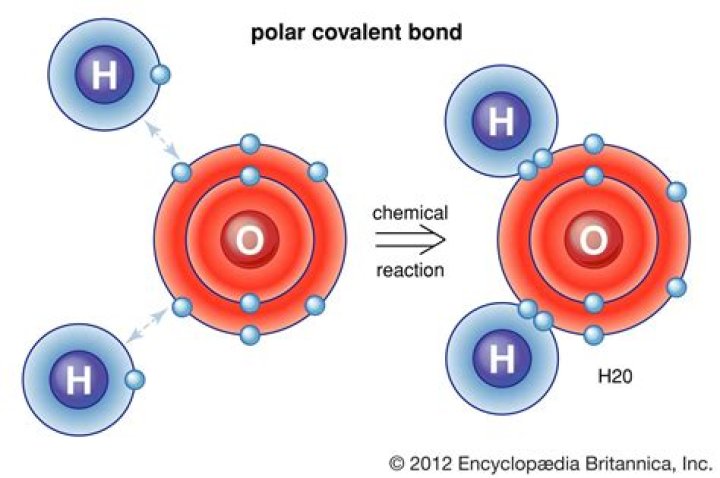
Strong linkages—called covalent bonds—hold together the hydrogen (white) and oxygen (red) atoms of individual H2O molecules. Covalent bonds occur when two atoms—in this case oxygen and hydrogen—share electrons with each other.
Is water molecule single bond?
It is a single covalent bond where each hydrogen get one electron from Oxygen so as to achieve inertness of Helium having atomic number 2 and Oxygen get one one electron from each hydrogen for getting inertness of Neon.Is water molecule double covalent bond?
Water is a Polar Covalent Molecule The unequal sharing of electrons between the atoms and the unsymmetrical shape of the molecule means that a water molecule has two poles – a positive charge on the hydrogen pole (side) and a negative charge on the oxygen pole (side).
How many single bonds are in a water molecule?
Each water molecule can form two hydrogen bonds involving their hydrogen atoms plus two further hydrogen bonds utilizing the hydrogen atoms attached to neighboring water molecules.
How does a water molecule bond?
Water has an amazing ability to adhere (stick) to itself and to other substances. … In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules. The attraction between individual water molecules creates a bond known as a hydrogen bond.
What type of bonding occurs between water molecules quizlet?
A water molecule is formed by covalent bonds between an oxygen atom and two hydrogen atoms. The bond between hydrogen and oxygen involves unequal sharing of electron – it is a polar covalent bond.What are water molecules?
A water molecule consists of two atoms of hydrogen linked by covalent bonds to the same atom of oxygen. Atoms of oxygen are electronegative and attract the shared electrons in their covalent bonds.
What kind of bond holds two water molecules to each other quizlet?The type of bonding found in water molecules is hydrogen bonding. It has hydrogen in it, and is made of hydrogen and oxygen. Another component is heat capacity, it uses the energy to break hydrogen apart.
Article first time published onWhat kind of bonds can hold two separate water molecules together?
The bonds that keep a water molecule together as a molecule are covalent bonds between the oxygen atom and the two hydrogen atoms.
Does water have ionic bonds?
Likewise, a water molecule is ionic in nature, but the bond is called covalent, with two hydrogen atoms both situating themselves with their positive charge on one side of the oxygen atom, which has a negative charge.
What is the angle of a water molecule?
A water molecule consists of two hydrogen atoms and one oxygen atom. The three atoms make an angle; the H-O-H angle is approximately 104.5 degrees. The center of each hydrogen atom is approximately 0.0957 nm from the center of the oxygen atom.
When a water molecule forms a hydrogen bond with another water molecule which of the following atoms are involved?
Two hydrogen atom from one molecule and one oxygen from the other molecule.
How do you find the water molecule?
Water consists of tiny particles called molecules. You can’t see them, not even under a microscope, they are much smaller than that. But even each water molecule consists again of smaller particles called atoms. Each water molecule consists of three: one oxygen atom (called O) and two hydrogen atoms (called H).
Why do water molecules have partial charges?
In the covalent bond between oxygen and hydrogen, the oxygen atom attracts electrons a bit more strongly than the hydrogen atoms. The unequal sharing of electrons gives the water molecule a slight negative charge near its oxygen atom and a slight positive charge near its hydrogen atoms.
What type of attraction occurs when water molecules attract other types of molecules?
10. Water molecules attracting other types of molecules is called adhesion. 11. Adhesion and cohesion together enable water molecules to move upward through narrow tubes against the force of gravity.
Why do hydrogen bonds form between water molecules?
In water molecules the oxygen atom attracts the negatively charged electrons more strongly than the hydrogen. This gives water an asymmetrical distribution of charge so that it is a polar molecule. … Because the water molecules are small, many of them can surround one molecule of the solute and form hydrogen bonds.
Which kind of bond holds atoms in water molecules together what attracts water molecules to one another?
The atoms in a water molecule are held together by polar covalent bonds. Water molecules are attracted to one another by hydrogen bonds.
Is a water molecule ionic or covalent?
Water is said to belong to the covalent category. In other words, water is made up of covalent bonds and not ionic bonds. If we look at the structure of water, then we will notice that the hydrogen atoms tend to share electrons with the oxygen atom making it covalent.
Is H2O ionic or molecule?
H2O is a covalent molecule. It’s also polar molecule. polar molecule occur when there is electronegativity difference (0.5 to 2.0) between bonded atoms. The Hydrogen and Oxygen respective electronegatives of 3.44 and 2.20.
Why is there a bond angle in water?
It’s because the two nonbonding 2p orbitals on oxygen impose a spatial repulsion, bending together the two hydrogens and decreasing the bond angle, which is measured such that it is ≤180∘ . (It is acceptable to just say 104.5∘ , but it is known to more decimal places than just one.)
What molecule can form hydrogen bond with water?
elementelectronegativity valueH2.1N3.0O3.5F4.1