How do you find the electron configuration for silver

Lily Fisher
Published Feb 17, 2026
To calculate an electron configuration, divide the periodic table into sections to represent the atomic orbitals, the regions where electrons are contained. Groups one and two are the s-block, three through 12 represent the d-block, 13 to 18 are the p-block and the two rows at the bottom are the f-block.
Why is Silver's electron configuration different?
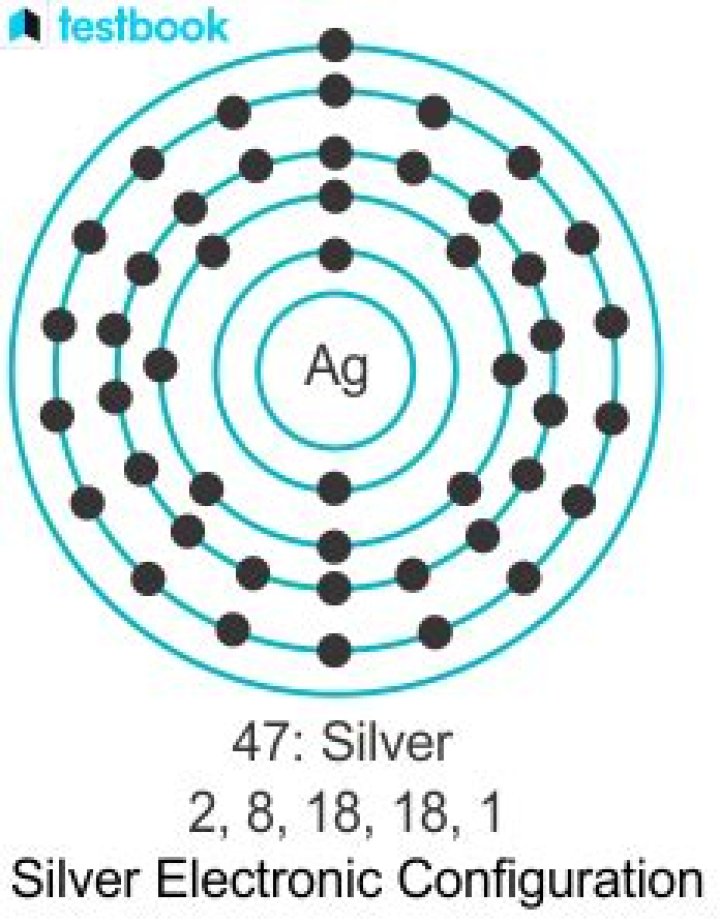
Silver, Ag , is located in period 5, group 11 of the periodic table, and has an atomic number equal to 47 . … The thing to remember here is that in silver’s case, the 4d orbitals will be completely filled. That implies that you won’t have two electrons in the 5s orbital, since one will be kept in the lower 4d orbitals.
What is e9 class configuration?
Electronic configuration is defined as the distribution of electrons into the orbitals of an atom. Every neutral atom consists of a fixed number of electrons which is equal to the number of protons and is called the atomic number.
What is the configuration of metals?
ELECTRONIC CONFIGURATION OF METALS AND NON – METALS Metals are elements that give off “free” electrons. Metals generally have anywhere between 1 to 3 electrons excess of a closed shell. Non-metals on the other hand have similar number of electrons in deficit of the numbers required for a closed shell.What is electronic configuration of Ag+?
Explanation: The ground state electron configuration of Ag is [Kr]4d105s1 . In order to form the Ag+ , an electron would be removed from the 5s sublevel, So the electron configuration of a Ag+ ion would be [Kr]4d10 .
How do you find the noble gas configuration for silver?
For silver , Z=47 . It is thus 11 protons removed from the last Noble Gas, which is krypton , Z=36 . And thus the electronic configuration of silver metal is: [Kr]4d105s1 .
What is silver's Valency?
The most common valency of silver metal is 1. The symbol of silver is Ag.
What is 1s 2s 2p 3s 3p?
1s 2s 2p 3s 3p represents the electron orbital energy levels.What is the electron configuration 1s2 2s2 2p6?
ABCarbon1s2 2s2 2p4Nitrogen1s2 2s2 2p5Silicon1s2 2s2 2p6 3s2 3p2Phosphorous1s2 2s2 2p6 3s2 3p3
What is the electronic configuration of the first 20 elements?Atomic NumberName of the ElementElectronic Configuration18Argon (Ar)[Ne] 3s2 3p619Potassium (K)[Ar] 4s120Calcium (Ca)[Ar] 4s221Scandium (Sc)[Ar] 3d1 4s2
Article first time published onWhat is the number of electrons in silver?
Number of Protons/Electrons: 47.
What is silver formula?
Silver is a metallic element with atomic symbol Ag, atomic number 47, and atomic weight 107.8682.
What is the symbol and valency of silver?
Therefore, the symbol of silver is Ag and the most common valency of silver metal is 1 and 3 .
Does silver show variable valency?
silver can sometimes lose more electrons than are present in its valence shell i.e. loss from the penultimate shell and hence exhibit more than 1 or variable valency.
What element is 1s2 2s2 2p6 3s2 3p6 4s2 3d10?
ABcalcium1s2 2s2 2p6 3s2 3p6 4s2chromium1s2 2s2 2p6 3s2 3p6 4s1 3d5 !copper1s2 2s2 2p6 3s2 3p6 4s1 3d 10 !bromine1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p5
Which element has the electron configuration of 1s 2 2s 2 2p 6 3s 2 3p 4?
ElementAtomic numberElectron configurationsilicon141s22s22p63s23p2phosphorus151s22s22p63s23p3sulfur161s22s22p63s23p4chlorine171s22s22p63s23p5
What is the electron configuration order?
Aufbau Principle According to this principle, electrons are filled in the following order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p…
Where is 5f on the periodic table?
In order to make the overall shape of the table more compact and convenient to display, scientists have adopted the convention of removing the elements with atomic number 57 through 70 and 89 through 102 (the latter being the 5f portion of the table) from their natural position between the s and d blocks and placing …
What does the 1 represent in electron configuration?
Electron configuration notation eliminates the boxes and arrows of orbital filling diagrams. Each occupied sublevel designation is written followed by a superscript that is the number of electrons in that sublevel. For example, the hydrogen configuration is 1s1, while the helium configuration is 1s2.