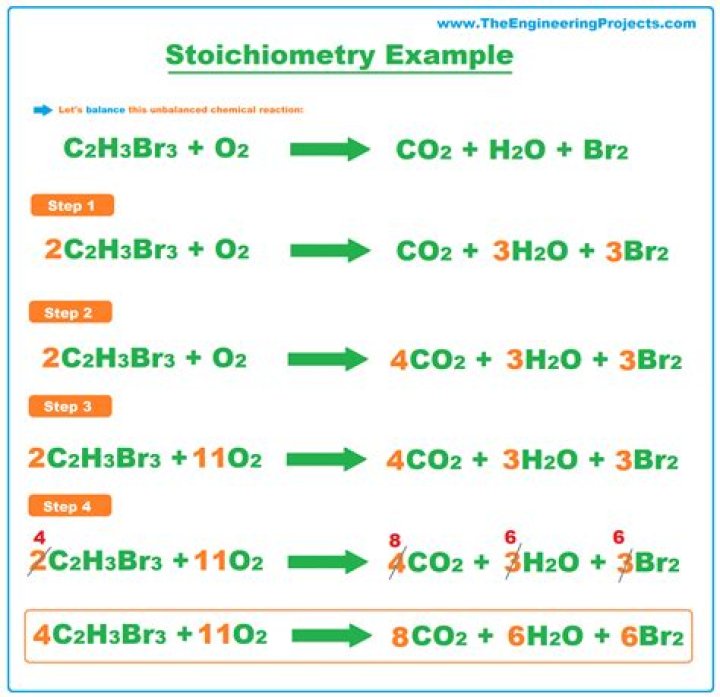
How do you do stoichiometry in chemistry

Sarah Rodriguez
Published Feb 28, 2026
Write the balanced chemical equation.Convert the units of the given substance (A) to moles.Use the mole ratio to calculate the moles of wanted substance (B).Convert moles of the wanted substance to the desired units.
What is stoichiometric formula?
The stoichiometry of a balanced chemical equation identifies the maximum amount of product that can be obtained. The stoichiometry of a reaction describes the relative amounts of reactants and products in a balanced chemical equation.
What does stoichiometry mean in chemistry?
In chemistry it is very important to understand the relationship between reactants and products in a reaction. Stoichiometry is exactly that. It is the quantitative relation between the number of moles (and therefore mass) of various products and reactants in a chemical reaction.
Is stoichiometry easy?
Stoichiometry can be difficult because it builds upon a number of individual skills. To be successful you must master the skills and learn how to plan your problem solving strategy. Master each of these skills before moving on: Calculating Molar Mass.Where is stoichiometry used?
Stoichiometry is at the heart of the production of many things you use in your daily life. Soap, tires, fertilizer, gasoline, deodorant, and chocolate bars are just a few commodities you use that are chemically engineered, or produced through chemical reactions.
How will the stoichiometric point be determined in this experiment?
This stoichiometric point can be determined by plotting the amount of precipitate formed vs. the amount of A used in the experiments, as in Figure 2-1.
What is stoichiometry in chemistry Quora?
In chemistry, stoichiometry is a kind of math that helps you measure and calculate amounts of chemicals. There are several quantity units used in stoichiometry. Some of them, like grams and liters, will be familiar even to people who have not studied chemistry.
How do chemical engineers use stoichiometry?
Stoichiometry is the mathematical understanding of how two materials relate to each other and the specifics of their reactions used to increase knowledge. … In order to solve this problem chemical engineers will have to work with chemicals and biological material to create lasting therapies for people.Why is stoichiometry important in chemistry?
Why Stoichiometry Is Important You can’t understand chemistry without grasping the basics of stoichiometry because it helps you predict how much of a reactant participates in a chemical reaction, how much product you’ll get, and how much reactant might be left over.
What is stoichiometry of solutions?Stoichiometry allows us to work in solution by giving us the concept of solution concentration, or molarity. … It is defined as the moles of a substance contained in one liter of solution. For instance, if a solution has a concentration of 1.20 M NaCl, this means that there are 1.20 moles of NaCl per liter of solution.
Article first time published onWhat does ecological stoichiometry explain?
Ecological stoichiometry seeks to discover how the chemical content of organisms shapes their ecology. Ecological stoichiometry has been applied to studies of nutrient recycling, resource competition, animal growth, and nutrient limitation patterns in whole ecosystems.
What is an example of stoichiometry?
Stoichiometry is the field of chemistry that is concerned with the relative quantities of reactants and products in chemical reactions. … For example, when oxygen and hydrogen react to produce water, one mole of oxygen reacts with two moles of hydrogen to produce two moles of water.
How is stoichiometry used to determine the concentration of an unknown solution?
Determining the Concentration of an Unknown Solution Using a Titration. … The volume of titrant added, its concentration, and the coefficients from the balanced chemical equation for the reaction allow us to calculate the total number of moles of the unknown in the original solution.
How is stoichiometry used in titration?
In order to perform an acid-base titration, you must have a solution of acid or base with a known concentration. … Based on the stoichiometry of your neutralization reaction, you then know how many moles of acid or base were in the unknown sample.
What is stoichiometry in titration?
Titration: Experiment which determines the concentration of a solute (reactant) using its reaction of known stoichiometry with another solution (reactant) of known concentration. … In a titration the titrant is added dropwise until the reaction is complete.
Why is reaction stoichiometry important give some examples?
Explanation: Stoichiometry allows us to make predictions about the outcomes of chemical reactions. … Predict the mass of a product of a chemical reaction if given the starting masses of reactants. Predict the volume of a gas which will be produced by a reaction if given the starting amounts of reactants.
How is stoichiometry used in pharmaceuticals?
Nurses routinely use stoichiometry to convert the drip ratios of medication to the prescribed dosages. They use a persons weight and conversion factors to determine the correct dosage of medicine to give. If the stoichiometry calculations are done incorrectly the patient can potentially be harmed.
How is stoichiometry used in real life?
Stoichiometry continues to be useful in many walks of life—a farmer determining how much fertilizer to use, figuring out how fast you have to go to get somewhere in a specific period of time, or just to make conversions between systems like Celsius and Fahrenheit.
How is stoichiometry used in tires?
What is stoichiometry? … Stoichiometry, in its simplest definition, merely refers to the relative proportion of components. For example, the stoichiometric relationship of steering wheels to tires in a normal automobile is 1:4.
How Stoichiometry is essential in food industry?
Stoichiometry is used in industry quite often to determine the amount of materials required to produce the desired amount of products in a given useful equation.
When studying ecological stoichiometry the ratio of which three elements will be most in focus?
Since the days of Redfield (1958), stoichiometric approaches have had a predominant focus on three elements: C, N, and P.
What is the growth rate hypothesis?
The growth rate hypothesis predicts that organisms with higher maximum growth rates will also have higher body percent phosphorus (P) due to the increased demand for ribosomal RNA production needed to sustain rapid growth.