How is sodium polyacrylate Formed

Ava Hall
Published Mar 06, 2026
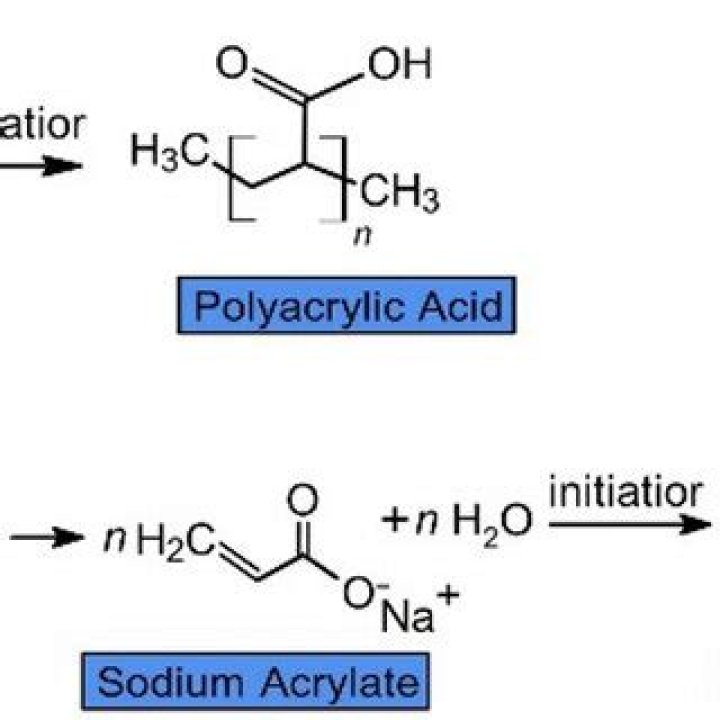
Sodium polyacrylate is produced by the reaction between acrylic acid (H2C=CHCOOH) and its sodium salt (H2C=CHCOONa). The product of this reaction is a long-chain copolymer consisting of alternate units of acrylic acid and sodium acrylate.
What elements make up sodium polyacrylate?
Sodium polyacrylate is the absorbent material found in baby diapers, artificial decorative snow, and detergents. They polymer structure is made up of one molecule of oxygen, one molecule of sodium oxide, a carbon base, and three hydrogen molecules.
Is sodium polyacrylate natural or synthetic?
What Is Sodium Polyacrylate? Sodium polyacrylate is a synthetic polymer used in cosmetics and skincare products. It is used to improve the sensory feel and stability of products. It functions as a thickening agent, texture enhancer, film-forming agent, and emulsion stabilizer.
How do you make sodium polyacrylate and water?
- Place 1/8 teaspoon of sodium polyacrylate powder in a plastic cup.
- Place 1/4 teaspoon of PolySnow™ in another cup.
- Using a 4 oz. …
- Continue adding water to both cups until absorption seems to stop.
Is sodium polyacrylate a Microplastic?
Sodium polyacrylate is not considered to be a plastic. … The family of polymers is a large one and includes materials other than plastics and resins. It is more often categorised as a superabsorbent material, it can absorb many times its own weight of water and hold it in creating a soft stable gel.
How do you make sodium polyacrylate?
- Add a small scoop (about a teaspoon) of SPA to the Erlenmeyer flask using the powder scoop.
- Add 25 ml of water to the SPA in the flask.
- Wait until all of the water is absorbed. This step won’t take long.
- Turn over the flask in your hand and hold the “snow.” It will be cool to the touch.
What is super absorbent polymer made of?
Superabsorbent polymers (SAP) are now commonly made from the polymerization of acrylic acid blended with sodium hydroxide in the presence of an initiator to form a poly-acrylic acid, sodium salt (sometimes referred to as cross-linked sodium polyacrylate).
What is another name for sodium polyacrylate?
Sodium polyacrylate, also known as waterlock, is a sodium salt of polyacrylic acid with the chemical formula [−CH2−CH(CO2Na)−]n and has broad applications in consumer products. This super-absorbent polymer (SAP) has the ability to absorb 100 to 1000 times its mass in water.What turns water into a gel?
As seen on the Ellen DeGeneres show, and also known as slush powder, Water Gel turns from a powder to a gel in just seconds. Sodium polyacrylate absorbs from 500-800 times its weight in water and is actually the secret ingredient that’s used to absorb “liquid” in baby diapers!
Who invented sodium polyacrylate?Robert Niles Bashaw, Bobby Leroy Atkins, and Billy Gene Harper are considered the first inventors of sodium polyacrylate.
Article first time published onCan you eat sodium polyacrylate?
This polymer is highly toxic when inhaled or ingested, causing damage to your eyes, skin and lungs. If you have accidentally inhaled or ingested sodium polyacrylate, seek medical attention immediately.
How do you make fake snow with sodium polyacrylate?
All you need to do to make this type of fake snow is add water to the sodium polyacrylate. Add some water, mix the gel. Add more water until you have the desired amount of wetness. The gel will not dissolve.
Does sodium polyacrylate have polar bonds?
The most popular superabsorbent polymer is a powder called Sodium Polyacrylate. … The Sodium Polyacrylate polymer, shown below in blue, contains a polar charge that allows it to combine with other liquids by forming hydrogen bonds.
Can sodium polyacrylate be recycled?
The sodium polyacrylate manufacturer, Recyc PHP, recycles a super absorbent polymer (SAP or AGM). It’s reclaimed from post-industrial rejects by Recyc PHP. This very pure recycled material has the following characteristics: It rapidly absorbs water-based liquids.
Is sodium polyacrylate ionic or covalent?
Sodium polyacrylate is a polymer (a long chain molecule) with ionic groups (negative charges with accompanying positively charged sodium ions) all along its length. It is these ionic groups that allow it to absorb a huge amount of water.
How do you identify cosmetic microplastics?
Infrared (IR) Spectroscopy An IR microscope is capable of detecting and identifying microplastics measuring as small as a few micrometers.
Why are microplastics used in cosmetics?
Microplastics are typically used to add exfoliating properties to a skincare products. They can help remove dry, dead skin cells and unclog pores which is why they’re often found in peelings and other exfoliating products such as bath gels.
Are acrylates microplastics?
Acrylates copolymer is a well-established microplastic. It does not just disappear once washed down the drain, and wastewater systems cannot remove the plastic particles during water treatment. Fish are ingesting the microplastic particles, but humans eat them too.
How do you make super absorbent polymers?
Superabsorbent polymers are now commonly made from the polymerization of acrylic acid blended with sodium hydroxide in the presence of an initiator to form a poly-acrylic acid sodium salt (sometimes referred to as sodium polyacrylate). This polymer is the most common type of SAP made in the world today.
Is super absorbent polymer safe?
The safety of superabsorbent polymers Superabsorbents are safe for use in absorbent hygiene products. They have been extensively tested and researched. Scientists and doctors alike have reviewed the research and confirm that superabsorbent material is safe.
What dissolves sodium polyacrylate?
Sodium polyacrylate is soluble in water. … This is driven by the dissociation of the sodium carboxylate salt in the water and the hydrogen bonding interactions between the water and the polymer.
How is gel made?
A simple gel can be made using cornstarch and water, and varying the ratio will change its properties. … When water is added and it is heated, some of the bonds between the glucose molecules break and the starch undergoes “gelatinization,” which means, you have yourself your very own, homemade gel!
What is snow powder made of?
Instant Snow Polymer is made by cross-linking molecules of tile sodium polyacrylate polymer, the material that gives disposable diapers their super absorbent quality. These small individual cross-linked clusters appear as a white powdery substance.
Can you make water into powder?
Dry water actually consists of 95% liquid water, but the silica coating prevents the water droplets from combining and turning back into a bulk liquid. … The result is a white powder that looks very similar to table salt. It is also more commonly known among researchers as empty water.
How do you make water based gel at home?
- Dissolve 1/2 teaspoon of gelatin in warm water.
- Add more gelatin as needed to reach the desired consistency, and stir.
- Cool your hair gel in the refrigerator for about three hours, or until set.
- Add a few drops of your favorite essential oil, if you’d like, and mix well.
How do you make a hydrogel?
- Measure out ¼ cup cold deionized water into mixing bowl.
- Sprinkle 1 Tbsp of gelatin over the cold water and let hydrate for at least 1 minute.
- Add tea bag and glycerin to measuring cup.
- Boil and pour ¾ cup deionized water over the tea bag and glycerin.
- Let steep until tea is desired strength and color.
Can sodium polyacrylate go down the drain?
If you have accidentally inhaled or ingested sodium polyacrylate, seek medical attention immediately. If it enters sewer or drainage systems in large quantities, it can cause serious clogging and should be dealt with immediately. Dispose of excess powder and/or gelled material in the trash. Do not put down the sink!
What are 3 uses for sodium polyacrylate?
Sodium polyacrylate is an absorbent polymer that is used as an emulsion stabilizer, a hair fixative, a film former, skin conditioner, and a viscosity agent [1].
How much sodium polyacrylate is in a diaper?
The Pampers diaper is composed of an average of 3.97 g of sodium polyacrylate, linked on a thin thread, and an average of 0.71 g of cotton. Huggies has an average of 1.88 g of fine powdery sodium polyacrylate and an average of 6.97 g of cotton.
When was sodium polyacrylate made?
Super-absorbent polymers, such as sodium poly(acrylate), were first developed in the late 1960s by the US Department of Agriculture. They were searching for ways to hold moisture in the soil more effectively.
Is polyacrylate polymer toxic?
Superabsorbent sodium polyacrylate polymeric hydrogels that retain large amounts of liquids are used in disposable diapers, sanitary napkins, and other applications. These polymers are generally considered “nontoxic” with acute oral median lethal doses (LD50) >5 g/kg.